
Diabetic foot ulcers (DFUs) are a common and severe complication of diabetes, affecting up to 15% of individuals with diabetes at some point in their lifetime. Although DFUs are not a direct cause of death, they often lead to infection and lower extremity amputation, which can have significant morbidity and mortality implications. The incidence of DFUs varies greatly depending on the population studied and the methods used to identify them.
Furthermore, the incidence is increasing due to the rise in the prevalence of diabetes, particularly in low- and middle-income countries where diabetes is becoming more common. Consequently, the burden of DFUs is expected to continue to rise in the coming years. In this article, diabetic foot ulcer stages, mechanisms of ulcer formation, and treatment options will be discussed.
How do we classify diabetic foot ulcers?
Several classification systems have been proposed in the past years to classify diabetic foot ulcers. However, none of them have been universally accepted.
Wagner Classification System
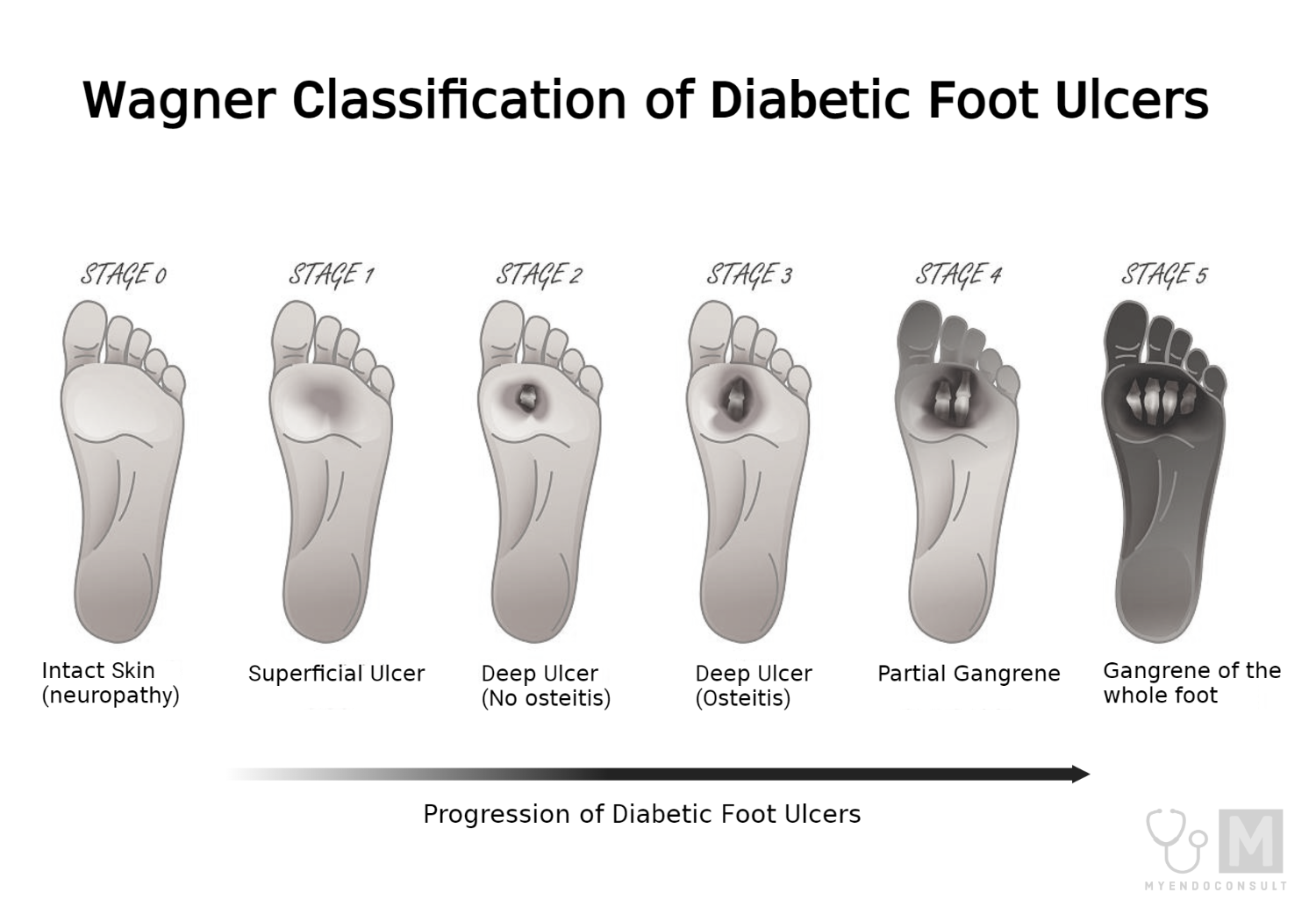
In the early 1980s, the Wagner-Meggitt grading system was established. It is the most widely recognized classification system that uses a series of grades from 0-5. The first four grades are based mainly on the depth of the wound, while grades 4 and 5 are determined by the severity of gangrene and decreased perfusion in the foot (1, 2). Grades 4 and 5 are limb-threatening grades which, in most instances, unfortunately, result in an amputation.
Table 1. Wagner Classification of Diabetic Foot Ulcers
| Grade | Description |
| Grade 0 | Inta, symptoms like pain in the foot (neuropathy) |
| Grade 1 | A superficial ulcer that involves the entire thickness of the skin but not the underlying tissues |
| Grade 2 | A deep ulcer that penetrates down to the muscle and Ligaments but does not affect the bones |
| Grade 3 | A deep ulcer that involves inflammation of the bone, Abscess formation |
| Grade 4 | Partial foot or localized gangrene |
| Grade 5 | Severe gangrene that extends to the whole foot |
However, Wagner’s grading system does not properly address the two most important factors, i.e., ischemia and infection. That’s why other classification systems were adapted, including the University of Texas classification system.
The University of Texas Classification System for Diabetic Foot Ulcers
The Texas system is advanced and classifies diabetic ulcers by wound depth (Grades 0 to 3) and then stages them (A to D) guided by the degree of ischemia and infection. It is frequently used in many clinical trials and diabetic foot centers and is better than the Wagner method in predicting wound healing.
Classification based on Grading (How deep is the ulcer?)
- Grade 0 : Pre- or post-ulcerative lesion that has healed.
- Grade 1 : Superficial wound that does not penetrate to tendon, capsule, or bone
- Grade 2 : Wound penetrates to tendon and capsule but no involvement of bone
- Grade 3 : Wound penetrating to bone or into a joint.
Classification based on Staging (is there evidence of ischemia or infection?)
- Stage A: clean wounds, i.e., no infection or ischemia
- Stage B: no ischemic infected wounds
- Stage C: ischemic wounds, non-infected
- Stage D: infected ischemic wounds
Other Classification Systems
Another classification system named PEDIS was designed by the International Working Group on the Diabetic Foot. This system uses four grades (1-4) and grades the wound based on five characteristics:
- Perfusion (blood supply to the area)
- Extent (size)
- Depth (tissue loss)
- Infection
- Sensation (neuropathy)
Similarly, the SAD classification system assesses the size, depth of ulcer, presence of bacterial infection or sepsis, arteriopathy, and denervation and grades these five clinical features on a 4-point scale (0–3). This system is relatively easy to understand and useful as a teaching tool.
Finally, diabetic foot ulcer is classified into three main categories: mild (involvement of only skin and subcutaneous tissues), moderate (affecting deeper tissues), and severe (associated with systemic signs of infection or metabolic instability).
Depending on how vascular disease and peripheral neuropathy affect the ulcer’s etiology, diabetic foot ulcers can be classified as neuropathic, ischemic, or neuroischemic in a simplified clinical classification method (3, 4).
Mechanism of Diabetic Foot ulcers
The etiology of diabetic foot ulcers is a complex and multifactorial process that involves various risk factors. These risk factors can be broadly classified into three groups: pathophysiologic changes, anatomic deformities, and trauma. Pathophysiologic alterations at the molecular level lead to peripheral neuropathy, peripheral vascular disease, and a weakened immune system, which ultimately impairs wound healing.
Peripheral neuropathy, characterized by loss of protective sensation and biomechanical abnormalities, is associated with almost 85% of diabetic foot ulcers (5). Also, the peripheral nervous system provides early warning signs, and damage to these indicators leads to inappropriate stress responses (6).
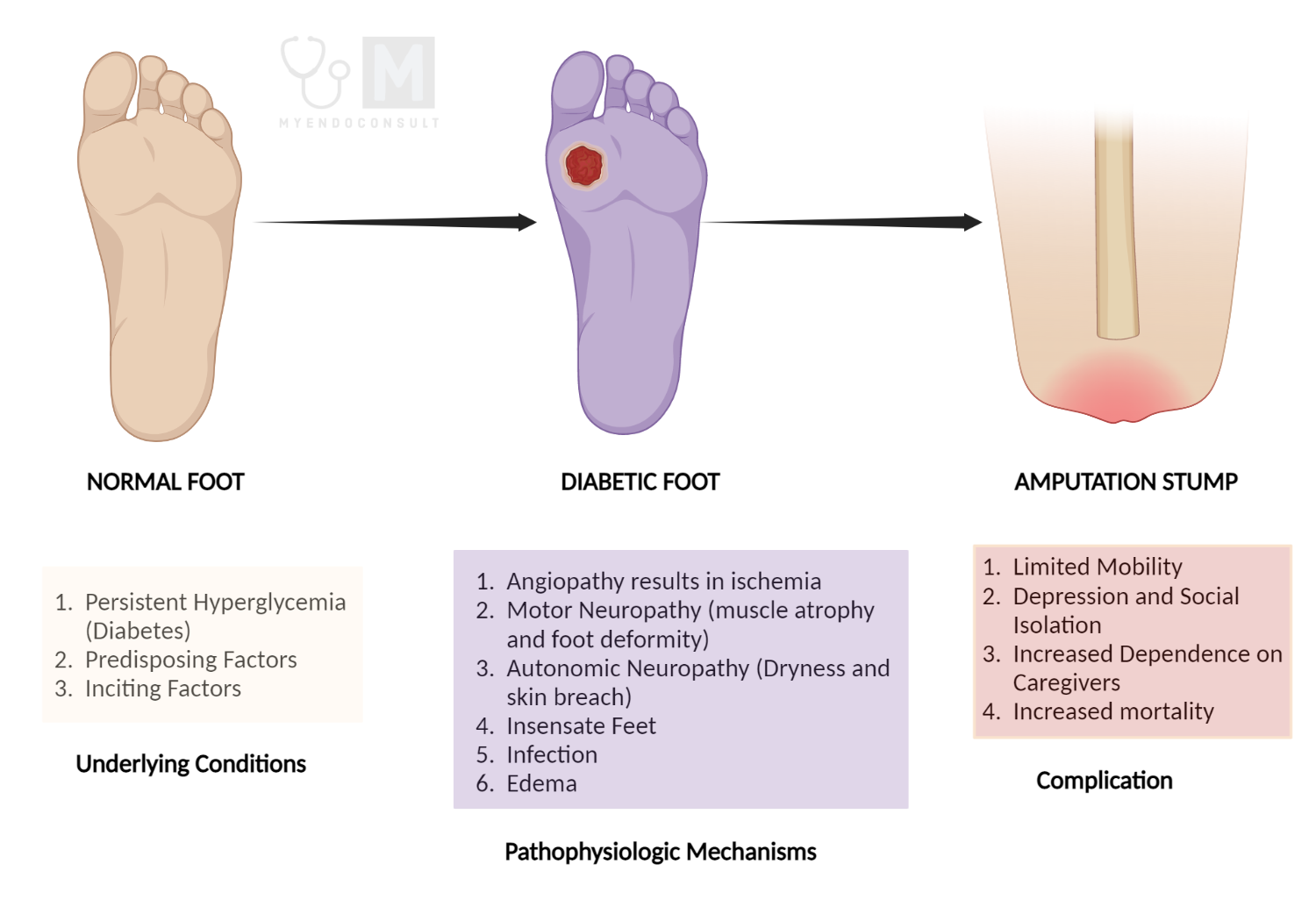
Anatomic foot deformities, including Charcot neuroarthropathy and motor neuropathy, raise peak plantar pressures and skin breakdown, leading to ulceration. Furthermore, extrinsic factors, such as acute or chronic trauma, also contribute to the development of diabetic foot ulcers. A long-standing history of diabetes, previous foot ulceration, and amputation are additonal risk factors for DFUs.
The other significant risk factors include a long-term history of diabetes, history of foot ulceration, and amputation.
Growth factors and cytokines are also fundamental for regulating the molecular mechanisms that promote cutaneous wound healing. Consequently, genetic mutations such as single nucleotide polymorphisms (SNPs) can significantly impact the variation of these growth factors and cytokines, thus predisposing some patients to diabetic foot ulcers.
It is crucial to understand that all of these factors generally occur in combination, significantly raising the risk of ulceration. To summarize, an insensate foot is frequently the first step on the pathway of diabetic foot ulceration. Then either acute or chronic trauma often occurs in an area with elevated peak plantar pressure, resulting in skin disintegration. Finally, impaired wound healing due to the reduced blood supply to the area and a deficiency in growth factors can all contribute to foot ulcer development (7,8).
A better understanding of these mechanisms will make it possible to develop targeted therapies tailored to each patient.
Treatment of Diabetic Foot ulcers
It is a well-known fact that the various factors involved in the pathogenesis of diabetic foot ulcers can differ in terms of the degree of severity of their effects. Hence, this could be one of the reasons why some treatments seem more effective for some patients than others.
The primary goal in treating diabetic foot ulcers is to obtain wound closure. The severity (grade) and vascularity of the foot ulcer and the presence of infection, all have a significant impact on its management.
Lower-grade ulcers are often treated with repeated debridement, pressure offloading, and wound dressings, while broad-spectrum antibiotics and occasionally limited or complete amputation is used to treat higher-grade wounds (9).
The non-operative management for wound care involves daily saline or similar dressings to maintain a moist wound environment while promoting exudate absorption around the ulcer site.
Also, pressure reduction at the wound site is another increasingly common method that helps to reduce ulceration in diabetic feet at risk. It can be achieved through offloading strategies, thus allowing the wound to heal. Various offloading devices include ‘total contact cast, assistive devices, total non-weight bearing, and custom fabricated shoes and insoles (10).
In cases of chronic osteomyelitis or soft tissue infection, pharmacological therapy with or without surgical intervention is needed. It has been demonstrated that a targeted antibiotic course based on the findings of wound cultures is beneficial for diabetic foot ulcers with concomitant infection. Depending on the severity, treatment could last from two weeks to two months.
Furthermore, for grade 3 or greater ulcers, surgical debridement is required. According to an evaluation of a 10-year review of standardized wound care protocol, diabetic amputation rates have tremendously decreased in patients through timely and effective debridement (11, 12).
Several studies suggest that optimal glycemic control is significant in slowing the progression of neuropathy (the cause of 90% of foot ulcers) in patients with diabetes mellitus.
Hyperbaric oxygen therapy (HBOT) is a promising adjunctive therapy for diabetic foot ulcers (DFUs) that are resistant to standard wound care management. It involves breathing 100% oxygen at elevated atmospheric pressure (usually at 1.4 atmosphere absolute) for a specified period. The benefits of HBOT include enhanced angiogenesis, improved bactericidal activity, and the promotion of wound healing by increasing tissue oxygenation.
Despite the promising results, there is still a lack of consensus on the use of HBOT in DFUs (13). In addition, HBOT is not without risks. It has been associated with barotrauma, oxygen toxicity, and complications related to using the hyperbaric chamber. Accordingly, while HBOT has shown promise in treating DFUs, further research is needed to establish its efficacy and safety before it can be routinely recommended for managing DFUs.
In order to prevent a recurrence, diabetic foot should be regarded as a life-long condition and treated accordingly with routine foot care.
Conclusion
Although the exact pathogenesis of DFUs is not fully understood, several factors contribute to their development, including peripheral neuropathy, peripheral arterial disease, foot deformities, trauma, and poor glycemic control. Several risk factors have been identified that increase the likelihood of developing DFUs, including a longer duration of diabetes, higher HbA1c levels, and a history of previous foot ulcers.
Effective management of DFUs requires a multidisciplinary approach and should include a focus on glycemic control, pressure offloading, and wound care. Furthermore, early detection and intervention are crucial in preventing complications and reducing the likelihood of amputation.
In conclusion, DFUs are a common and serious complication of diabetes that require prompt and effective management to prevent significant morbidity and mortality. Although their incidence is increasing, appropriate interventions can reduce the burden of DFUs and improve outcomes for individuals with diabetes.
Helpful Diabetic Foot Care Tips for patients
There are many steps that people with diabetes can take to care for their feet and prevent foot ulcers. Here are some detailed tips for diabetic foot care and prevention (patient education guide):
- Inspect your feet daily: Check your feet daily for any cuts, blisters, or other abnormalities. Use a mirror or ask a family member to check your feet if you have trouble seeing them.
- Wash your feet daily: Wash your feet daily with warm water and mild soap. Dry them thoroughly, especially between the toes.
- Moisturize your feet: Use a moisturizing cream or lotion to keep your feet from drying out. However, avoid putting lotion between your toes, as this can cause infection.
- Wear proper footwear: Always wear shoes and socks, even indoors. Choose shoes that fit well and have a wide and deep toe box to avoid pressure on your toes. Avoid shoes with high heels or pointed toes.
- Trim your toenails regularly: Trim your toenails straight across and file the edges. Don’t cut them too short or too rounded.
- Avoid going barefoot: Never walk barefoot, especially outdoors. Always wear shoes or slippers to protect your feet.
- Control your blood sugar: Keeping your blood sugar levels in a healthy range can help prevent nerve damage and other complications that lead to foot ulcers.
- Get regular foot check-ups: See your doctor or a podiatrist for a foot exam at least once a year or more often if you have foot problems.
- Quit smoking: Smoking can decrease blood flow to the feet, making it harder for foot injuries to heal.
- Stay active: Regular physical activity can improve circulation and help prevent foot ulcers.
In summary, taking good care of your feet is essential for people with diabetes. With these detailed tips for diabetic foot care and prevention, you can reduce your risk of foot ulcers and other complications.
References:
- Game F. Classification of diabetic foot ulcers. Diabetes/metabolism research and reviews. 2016;32:186-94.
- Levin ME, Bowker JH, Pfeifer MA. Levin and O’Neal’s the diabetic foot: Elsevier Health Sciences; 2008.
- Lavery LA, Armstrong DG, Harkless LB. Classification of diabetic foot wounds. The Journal of Foot and Ankle Surgery. 1996;35(6):528-31.
- Jeon BJ, Choi HJ, Kang JS, Tak MS, Park ES. Comparison of five systems of classification of diabetic foot ulcers and predictive factors for amputation. International wound journal. 2017;14(3):537-45.
- Bowling FL, Rashid ST, Boulton AJ. Preventing and treating foot complications associated with diabetes mellitus. Nature Reviews Endocrinology. 2015;11(10):606-16.
- Dinh T, Tecilazich F, Kafanas A, Doupis J, Gnardellis C, Leal E, et al. Mechanisms involved in the development and healing of diabetic foot ulceration. Diabetes. 2012;61(11):2937-47.
- Blakytny R, Jude EB. Altered molecular mechanisms of diabetic foot ulcers. The international journal of lower extremity wounds. 2009;8(2):95-104.
- Jhamb S, Vangaveti VN, Malabu UH. Genetic and molecular basis of diabetic foot ulcers: clinical review. Journal of Tissue Viability. 2016;25(4):229-36.
- Tiktin M, Celik S, Berard L. Understanding adherence to medications in type 2 diabetes care and clinical trials to overcome barriers: a narrative review. Current medical research and opinion. 2016;32(2):277-87.
- Hilton J, Williams D, Beuker B, Miller D, Harding K. Wound dressings in diabetic foot disease. Clinical Infectious Diseases. 2004;39(Supplement_2):S100-S3.
- Hsu C-R, Chang C-C, Chen Y-T, Lin W-N, Chen M-Y. Organization of wound healing services: the impact on lowering the diabetes foot amputation rate in a ten-year review and the importance of early debridement. Diabetes research and clinical practice. 2015;109(1):77-84.
- Tian X, Liang X, Song G, Zhao Y, Yang X. Maggot debridement therapy for the treatment of diabetic foot ulcers: a meta-analysis. Journal of wound care. 2013;22(9):462-9.
- Sharma, R., Sharma, S.K., Mudgal, S.K. et al. Efficacy of hyperbaric oxygen therapy for diabetic foot ulcer, a systematic review and meta-analysis of controlled clinical trials. Sci Rep 11, 2189 (2021).
Kindly Let Us Know If This Was helpful? Thank You!


