Hormones are a mysterious set of chemicals that flow throughout our bodies ferried by the blood and only exert their effects at specific sites expressing their complementary receptors.
These chemicals turn children into adults, control how we utilize energy, govern our appetite, and even affect our mood. In effect, hormones control everything in the human body. Interestingly, we have barely scratched the surface of our understanding of these chemicals.

Introduction to the history of hormones
We will start this historical journey in endocrinology by acknowledging the first person to use the word “hormone.” Ernest Henry Starling (1866-1927), a physiologist, discovered the hormone secretin and coined the term hormone during a collegial dinner at the University of Cambridge.
It is said that during a discussion with colleagues, he struggled to find the proper term for internal secretions that exert their effects at a distant site. The Greek word “hormao,” which means “I excite,” was proposed by an unknown colleague. The story goes that Starling used the previously unknown word, hormone, for the first time at a society meeting.
The four cardinal contributions by Ernest Henry Starling (1866-1927) include :
- Starling’s Principle (remember oncotic and hydrostatic pressures?)
- Discovery of Secretin (that gut hormone)
- Frank-Starling Law of the heart (effects of stretching and relaxation of cardiac muscle)
- Experimental evidence of the existence of anti-diuretic hormone
History of Testosterone
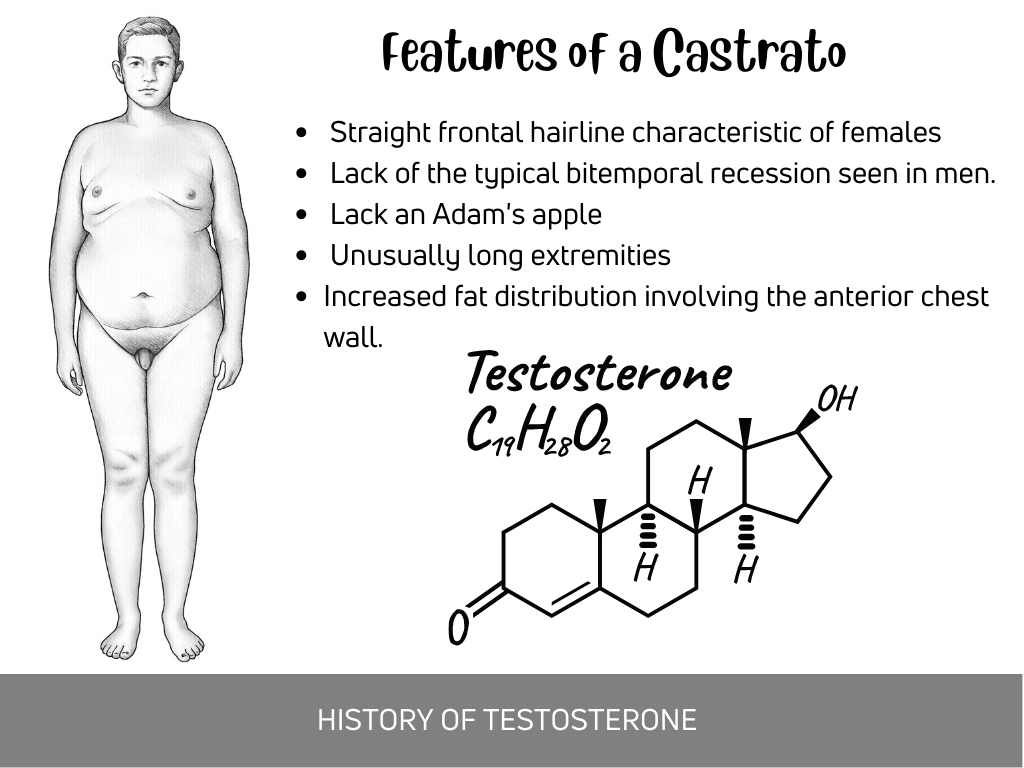
Carlo Maria Michelangelo Nicola Broschi, known by his stage name Farinelli, was an opera singer who lived in 18th century Italy. He had a high-pitched soprano voice range typically not exhibited by fully grown adult men. The reason for his remarkable voice was that he was a “castrato” – referring to a castrated man (“lacking testicular function”).

He was castrated before puberty and was never given a chance to break his voice during his teenage years. Unfortunately, he developed a unique set of clinical features consistent with male hypogonadism. For example, he had a straight frontal hairline characteristic of females and not the typical bitemporal recession seen in men. In addition, he lacked an Adam’s apple, had unusually long extremities, and increased fat distribution involving his anterior chest wall.
It is worth noting that humans have known about the effects of castration on animals for centuries, but we had no means of explaining its dramatic effects on the male species of humans and animals alike.

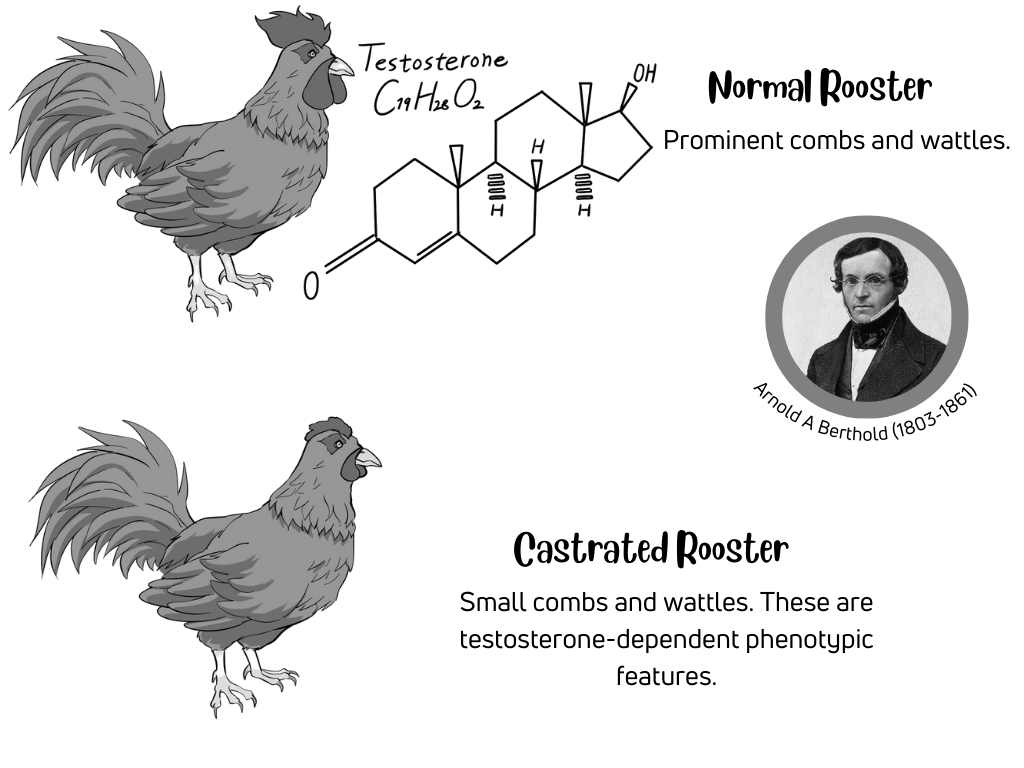
In the 1840s, Professor Arnold A Berthold (1803-1861) conducted a series of experiments on roosters. In his paper published in German in 1849, he outlined his systematic approach to observing the effects of castration and re-implantation of testes on typical male features of roosters.

In summary, by removing the testes of roosters (castration), he was able to show that the unfortunate roosters were no longer aggressive towards other males, had no interest in hens, and developed small combs and wattles (androgen-dependent phenotypic features). The roosters had typical male features after transplanting the testes to the abdominal cavity.
Berthold concluded that the testes produced a chemical ferried to various sites in the body to exert its effects. In essence, he was the first to describe the effects of testosterone.
Charles-Édouard Brown-Séquard, an eminent clinician known to medical trainees – “Brown-Séquard Syndrome” carried out a rather bizarre experiment in the 1800s. He prepared a rather nauseating concoction composed of “blood from the testicular vein…semen and testicular juice extract from a dog or guinea pig”.
He injected himself with this concoction in a scene that might seem illogical to modern clinicians. Being an eminent clinician of the day, he was allowed to present his findings at the academy of science in Paris, France. Brown-Séquard, then aged 72 years, reported experiencing an improvement in his physical strength and powers of concentration. Brown-Séquard’s extract became an overnight sensation worldwide with claims of being the new “Elixir of life.” It, however, goes without saying that this was most likely a placebo effect.
Indeed later evidence showed that to obtain an appreciable amount of testosterone from the testes, about one-quarter ton of a bull’s testes would be required. Organotherapy, the process of injecting the extracts of various glands, became a medical fad after Brown-Séquard’s experiment.
History of Thyroid Disease
The first successful treatment in endocrinology occurred in a patient with myxedema (severe hypothyroidism). Sir Victor Horsely, between the years 1884 and 1890, confirmed the relationship between a debilitating endocrine condition of the day, myxedema and the thyroid gland.
He was able to show in monkeys and dogs that removal of the thyroid gland resulted in symptoms suggestive of hypothyroidism. More importantly, the use of thyroid extracts produced a remarkable improvement in symptoms of myxedema.
Horsely initially proposed the transplantation of sheep’s thyroid into humans. Although this led to significant improvement in symptoms, the effects were short-lived and sometimes harmful.

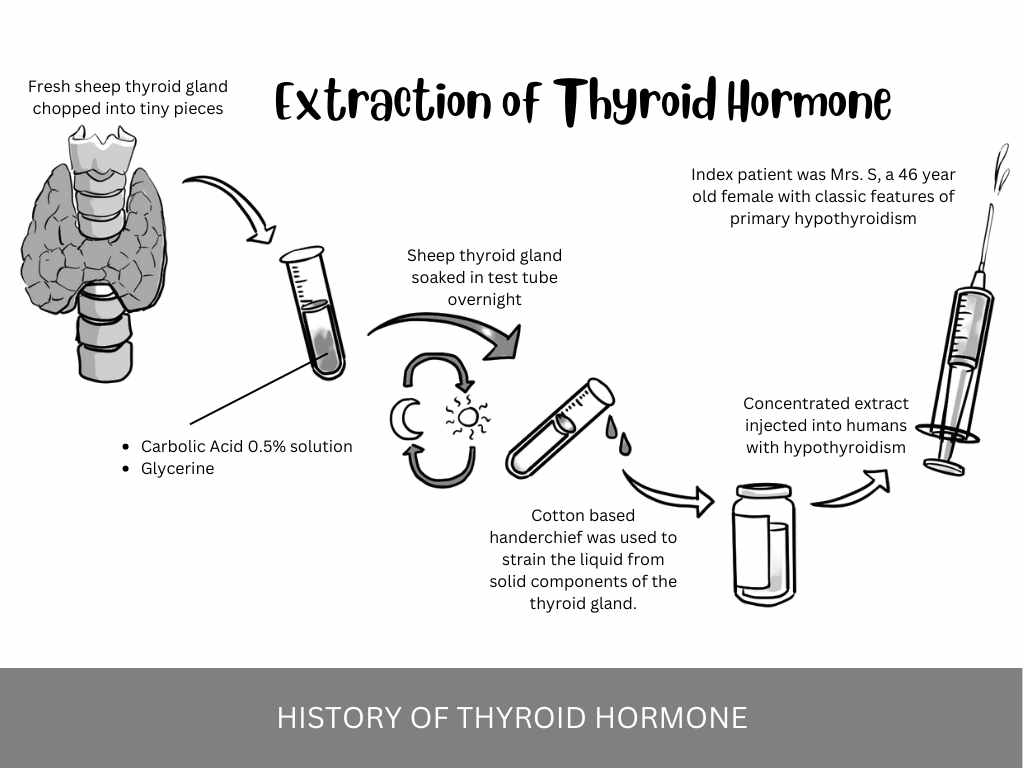
A student of Sir Horsely, George Murray extended his initial work. Murray used extracts from sheep’s thyroid in humans with myxedema.
These thyroid extracts were prepared by cutting up small pieces of the thyroid gland (obtained from the abattoir) storing them in carbolic acid overnight. He subsequently used a simple cotton-based handkerchief to strain the liquid component of thyroid tissue from its solid component. The extract was then injected subcutaneously, leading to rapid improvement in the symptoms of myxedema.
Murray published his report in the October 1891 issue of the British Medical Journal (BMJ). Although his findings were widely known at the time, Bettencourt and Serrano (Portuguese researchers) were the first to describe the use of subcutaneous thyroid extracts. It is worth noting that Murray cited this initial work in his BMJ report.
Selected figures
Hakaru Hashimoto, a graduate of Fukuoka Medical College (1903-1907). He reported a case series of a previously unknown form of thyroid disease. Four female patients aged 40 years and older underwent partial thyroidectomy and were found to have the characteristic histologic findings of chronic lymphocytic thyroiditis.
Hashimoto used the term “struma lymphomatosa” to describe the unique chronic inflammation of the thyroid gland1. Chronic lymphocytic thyroiditis is eponymously named “Hashimoto’s thyroiditis.” 2
Hashimoto compared his findings with those reported in Riedel’s thyroiditis. Patients with struma lymphomatosa, however, had less fibrosis noted on histology. Also, the thyroid gland of his subjects on clinical examination did not have the hard consistency of Riedel’s thyroiditis, which made it a distinct clinical entity3.
- Hashimoto’s discovery was mostly unrecognized for decades. His findings were again reported by another clinician who seemingly was unaware of Hashimoto’s original paper from 1912. He was eventually recognized at an international thyroid conference in 19383.
- His paper on struma lymphomatosa was initially published in the German language4.
Robert Graves
Robert Graves completed his medical education at the University of Dublin in 1818. He described exophthalmic goiter but was arguably not the first to describe this malady. Von Basedow and others documented the clinical features of diffuse toxic goiter before Robert Graves. Graves’ disease is also referred to as Basedow’s disease in some medical circles5.
- He described the “pin-point” pupillary findings in patients with a pontine hemorrhage.
- Graves advocated for the discontinuation of the practice of phlebotomy as a means of treating pyrexia.
- He proposed the concept of timing of peripheral pulses5.
History of Adrenaline
George Oliver used an arteriometer (a device for measuring the internal diameter of the radial artery) to evaluate a change in vascular flow in his child after injecting him with an extract of the suprarenal gland.
He reported observing an increase in the diameter of the radial artery after administering the extract. In an experiment by Schafer and Oliver, a glycerin-based extract of the suprarenal gland was injected directly into the vein of a dog after which they observed a dramatic increase in its blood pressure.
The duo carried out a second experiment on a cat and observed a significant increase in blood pressure after injecting the same extract. They reported their findings in March 1894 at the Physiological Society in a lecture titled “The physiological action of the extract of the suprarenal capsules.”
We now know that the series of experiments by George Oliver demonstrated the cardiovascular effects of the hormone adrenaline (or epinephrine, for our colleagues in North America).
Selected figures
Thomas Addison
Thomas Addison graduated from the University of Edinburgh medical school in 1815. “Addison’s anemia” was first reported in the London Medical Gazette in 1849 as ‘Anaemia – disease of the suprarenal capsules in which the disease is not distinctly separated from a new form of anemia.’ Trousseau (1801-1867) was the first to use the eponym “Addison’s disease” 6.
- Pernicious anemia is eponymously called Addison’s anemia7.
- The classical description of vitiligo is credited to Addison7.
Jerome W. Conn
Jerome W. Conn, a graduate of the University of Michigan Medical School (1929-1932). He made a lasting contribution to our understanding of the renin-angiotensin-aldosterone axis8. Primary hyperaldosteronism in the setting of a unilateral aldosterone-secreting tumor is widely reported as Conn’s syndrome9.
- Conn had an interest in diabetes pathophysiology, and his publications have provided insights into our understanding of insulin-resistant states10.
- Jerome Conn completed a year of training in general surgery, but redirected his interests to internal medicine and, subsequently, endocrinology10.
Edward Kendall
Edward Kendall completed his BSc, MSc, and Ph.D. degrees at Columbia University, New York City. He shared the 1950 Nobel Prize for Medicine and Physiology with Philip Hench (1896-1965), for the discovery of cortisone11.
- Kendall crystallized thyroxine in 1914, during his time at Mayo Clinic (Rochester, Minnesota)
- He isolated at least 28 adrenocortical hormones, including cortisone, a hormone he initially designated as “compound E” 11.
“Diabetes..ligate pancreatic duct of dogs…keep dogs alive till acini degenerate…leaving islets…try to isolate internal secretions…to relieve glycosuria”
History of Estrogen
There was a time in our shameful past as medical professionals when women were coerced into undergoing bilateral oophorectomy (removal of the ovaries) for hysteria, anorexia nervosa, and anxiety disorder. The central dogma of that day was that the nervous system controlled all parts of the human body.
Quite disconcertingly, the belief was that the ovaries were the nerve centers governing women’s behavior. This fueled the practice of ovarian removal and its unintended consequence of premature menopause. Over 100,000 young women underwent this barbaric procedure in Europe and subsequently developed severe vasomotor symptoms of menopause.
Josef von Halban (1870-1937) was a pioneer in hormone science. His seminal work on the internal secretions of the ovaries disproved the long-held belief that the ovaries were in direct communication with the nervous systems.
Halban showed that the ovaries exerted their systemic effects by releasing a special chemical (estrogen) directly into the bloodstream. He removed bits of the ovaries and uterus and transplanted these into the skin of guinea pigs. He was able to show that despite being anatomically displaced, the ovaries allowed an immature spayed guinea pig to attain normal puberty, confirming the role of a yet to be characterized hormone, we now know as estrogen.
Selected figures
Fuller Albright
Fuller Albright enrolled at Harvard Medical School in 1920. His research findings have been influential in our understanding of calcium and bone metabolism. He shares the eponymous term, McCune-Albright syndrome, with Donovan McCune.
He also made significant contributions to our understanding of osteoporosis, hyperparathyroidism, and Cushing’s disease12.
Albright had an initial desire to become an orthopedic surgeon but had to shelve those dreams because he did not have the dexterity required in this surgical subdiscipline. He was fascinated by calcium metabolism and subsequently turned his interests to the field of endocrinology12.
He investigated the role of estrogen in bone metabolism and postulated that estrogens stimulate osteoblast function and thus improve bone health12. Albright developed Parkinson’s disease when he was 36 years of age and, unfortunately, suffered an intracranial hemorrhage during a pallidotomy, a novel procedure at the time for Parkinson’s disease12.
History of Human Growth Hormone
Charles Byrne, also known as “The Irish Giant’ lived in the late 1700s and grew to a towering height of 8 feet 4 inches. He was a widely known “human curiosity” in London. John Hunter, a controversial scientific collector of the day, went against Byrne’s wish to be buried at sea and stole the young man’s mortal remains after his demise.
Byrne’s skeleton was subsequently displayed in a museum. Although this was unethical, it created the opportunity for Harvey Cushing, a luminary in the field of neurosurgery, to study the skeleton of this “scientific oddity.”
Cushing observed extensive erosion of the sella turcica (where the pituitary gland rests anatomically) and postulated that Byrne’s suffered from a malady of the pituitary gland. He was able to deduce that the chemical produced by the pituitary gland was responsible for growth.
Selected figures
Harvey Cushing
Harvey Cushing, a graduate of Harvard medical school (1891-1896). A neurosurgeon by training who became internationally recognized in the early 1900s. He made significant contributions to the field of neurosurgery and is known as the father of modern neurosurgery.
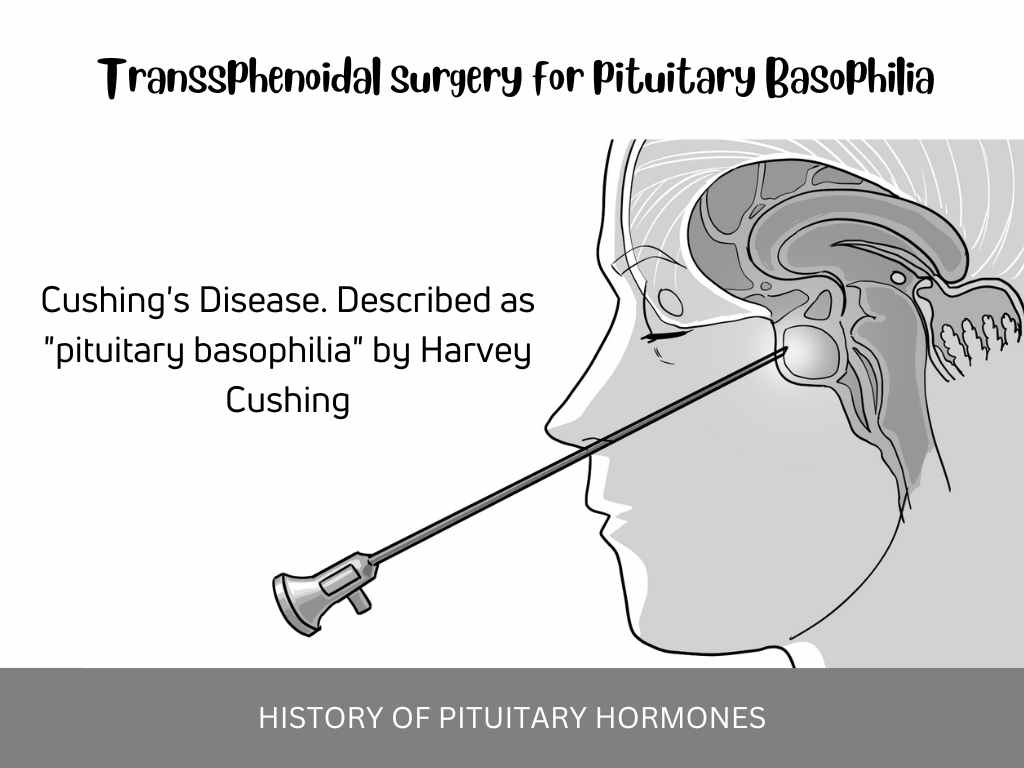
He, however, holds a special place in clinical endocrinology due to his description of “pituitary basophilia” or Cushing’s disease, a clinical syndrome eponymously named after him13.
- Harvey Cushing’s mentor was William Osler, a name known by many a medical trainee. They formed a professional and personal relationship during their time together at Johns Hopkins Hospital in Maryland. He wrote a two-volume biography of Osler in 1925.
- The anesthesia record, which details a patient’s vital signs during surgery, is credited to him.
- Other non-endocrine eponyms bearing his name include Cushing’s ulcers and Cushing’s reflex 14.
History of Insulin Therapy
Diabetes mellitus literally means “a sweet fountain,” which aptly describes the sweet taste of urine seen in patients with this condition. In the 19th century, it was irrevocably shown that removing the pancreas of dogs led to a canine equivalent of glycosuria and wasting syndrome consistent with diabetes mellitus.
The pancreas is composed of both an endocrine and exocrine component. Most of the gland is, however, composed of digestive enzymes, making the process of crude organotherapy (that is, extracting juices from a gland and administering it to a deficient subject) problematic.
Frederick Banting, a Canadian general practitioner, is credited with the discovery of insulin. He chanced upon an article that outlined an optimal approach to blunting the production of the digestive juices of the pancreas. This, in essence, would leave only the remaining pancreatic islet cells to make insulin, making it much easier to isolate this hormone. After pondering over this, Banting had found the most optimal approach to isolating this wonder chemical of the pancreas. These were his words, “Diabetes..ligate pancreatic duct of dogs…keep dogs alive till acini degenerate…leaving islets…try to isolate internal secretions…to relieve glycosuria”
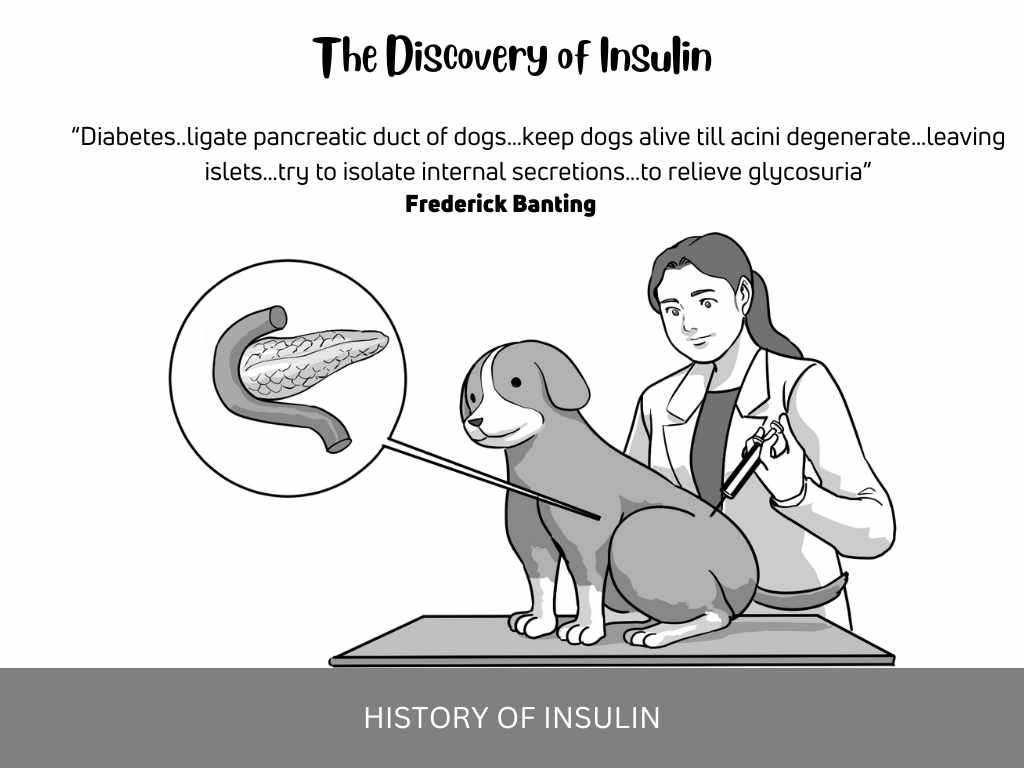
He used pancreatic extracts to cure dogs afflicted with surgically induced diabetes mellitus. Leonard Thompson was the first patient to receive the miracle healing of Banting’s pancreatic extract. He received the first dose of Banting’s canine pancreatic extract on 11th January 1922. Afterward, Thompson developed an allergic reaction to it.
As a result, Banting sought the assistance of James Collip in improving the purity of his extract. Thompson received a second injection of a more purified extract of this life-sustaining hormone 12 days later, after which he made a remarkable recovery with complete resolution of polyuria, polydipsia, wasting, and glycosuria. He, unfortunately, died at 26 years of age from pneumonia.
Insulin is now produced using recombinant DNA technology. This biosynthetic process involves the insertion of the coding sequence of insulin into a suitable host, in most cases Escherichia coli. The bacterium subsequently transcribes and translates the gene into the amino acid sequence of insulin. Thus, modern insulin is less likely to produce allergies than animal insulin.
Selected figures
Frederick Banting
Frederick Banting completed his medical education at the University of Toronto in 1916. He is known for the discovery of insulin, an honor he graciously shared with a junior colleague, Charles H. Best15.
- Banting and J.R.R Macleod were awarded the Nobel Prize in Medicine and Physiology in 192316.
- Leonard Thompson was the first patient to be treated with Banting’s insulin extract17.
History of the Parathyroid Glands
Richard Owen first described the parathyroids in a paper published in 1862. Owen, a professor of comparative anatomy in London, described the parathyroid gland as “a small yellow body attached to the thyroid” during an autopsy of a rhinoceros (18). However, the discovery of the parathyroid glands has been credited to Ivar Sandström, who published the discovery in 1880 in a paper titled “on a New Gland in Man and Several animals.” Due to the late anatomical description of these glands, they have often been referred to as “the last anatomic discovery.” 19
A simplified timeline of the discovery of hormones is available here.
References
- Loriaux DL. Hakaru Hashimoto (1881–1934). In: A Biographical History of Endocrinology. John Wiley & Sons, Ltd; 2016:269-273.
- Graham A, McCullagh EP. Atrophy and fibrosis associated with lymphoid tissue in the thyroid: struma lymphomatosa (Hashimoto). Arch Surg. 1931;22(4):548-567.
- Sawin CT. Hakaru Hashimoto (1881–1934) and his disease. The Endocrinologist. 2001;11(2):73-76.
- Volpé R. The Life of Dr. Hakaru Hashimoto. Autoimmunity. 1989;3(4):243-245.
- Jay V. Dr Robert James Graves. Arch Pathol Lab Med. 1999;123(4):284.
- Pearce JMS. Thomas Addison (1793-1860). J R Soc Med. 2004;97(6):297-300.
- Jay V. Thomas Addison. Arch Pathol Lab Med. 1999;123(3):190-190.
- Fajans SS. Jerome W. Conn, 1907-1994. Ann Intern Med. 1994;121(11):901.
- Christakis I, Livesey JA, Sadler GP, Mihai R. Laparoscopic Adrenalectomy for Conn’s Syndrome is Beneficial to Patients and is Cost Effective in England. J Investig Surg Off J Acad Surg Res. 2018;31(4):300-306.
- Loriaux D. Jerome W. Conn (1907–1994). The Endocrinologist. 2008;18(4):159-160.
- Shampo MA, Kyle RA. Edward C. Kendall—Nobel Laureate. Mayo Clin Proc. 2001;76(12):1188.
- Manring MM, Calhoun JH. Biographical sketch: Fuller Albright, MD 1900-1969. Clin Orthop. 2011;469(8):2092-2095. doi:10.1007/s11999-011-1831-0
- Perrins RJ. Harvey Cushing: A Life in Surgery. Soc Hist Med. 2006;19(3):576-578. doi:10.1093/shm/hkl074
- Loriaux DL. Harvey Williams Cushing (1869–1939). In: A Biographical History of Endocrinology. John Wiley & Sons, Ltd; 2016:202-206. doi:10.1002/9781119205791.ch48
- Tan SY, Merchant J. Frederick Banting (1891–1941): Discoverer of insulin. Singapore Med J. 2017;58(1):2-3.
- Bliss M. “Texts and Documents”: Banting’s, Best’s, and Collip’s Accounts of the Discovery of Insulin. Bull Hist Med. 1982;56(4):554.
- Karamanou M, Protogerou A, Tsoucalas G, Androutsos G, Poulakou-Rebelakou E. Milestones in the history of diabetes mellitus: The main contributors. World J Diabetes. 2016;7(1):1-7.
- Åkerström G, Hellman P, Hessman O, Segersten U, Westin G. Parathyroid Glands in Calcium Regulation and Human Disease. Ann N Y Acad Sci. 2005;1040(1):53-58.
- Johansson H. The Uppsala anatomist Ivar Sandström and the parathyroid gland. Ups J Med Sci. 2015;120(2):72-77.

