The mechanism of action of growth hormone and its clinical application in the management of adult growth hormone deficiency will be reviewed.
Physiology
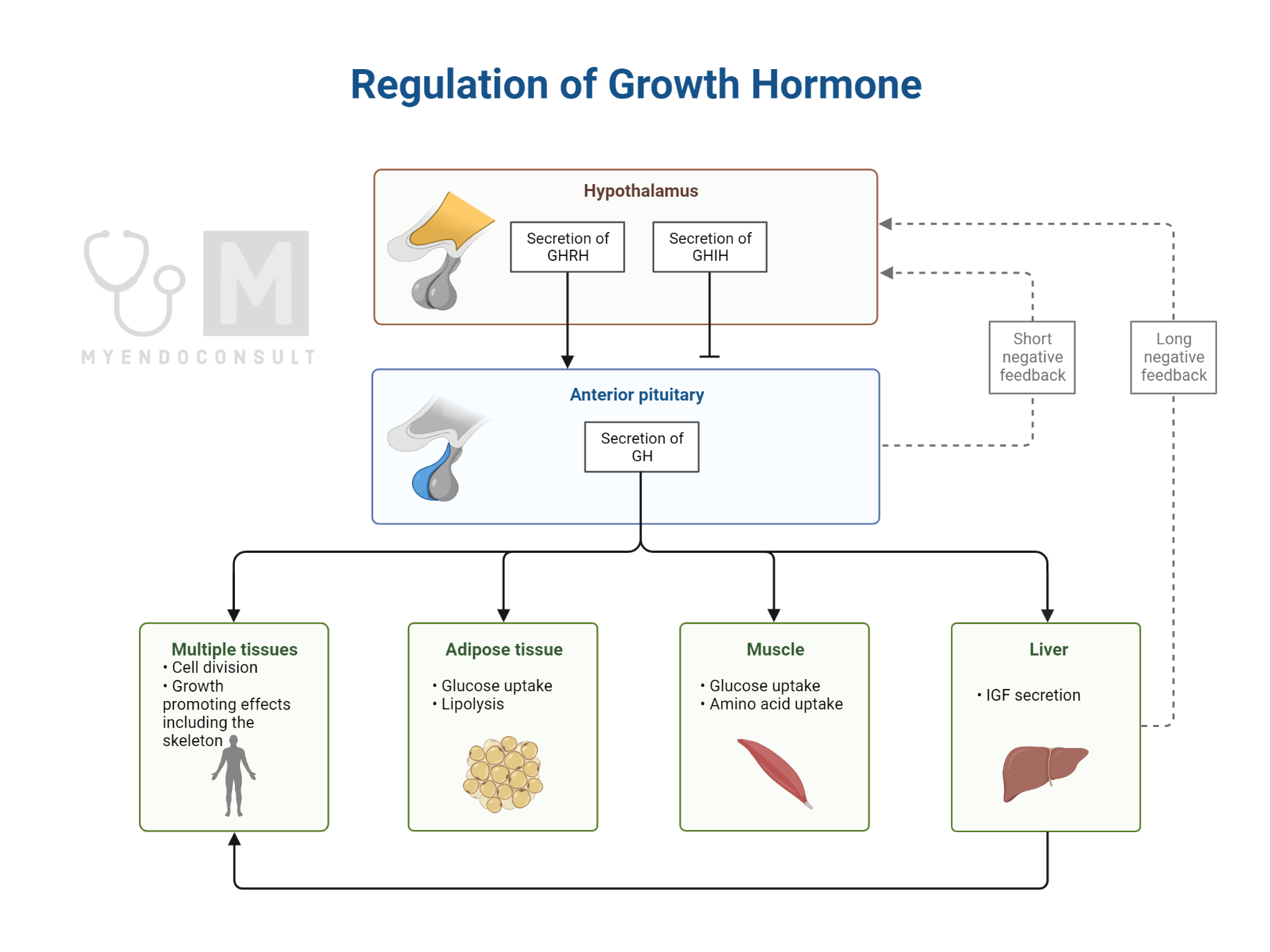
Anabolic effects of growth hormone: Basic growth hormone physiology was reviewed in another section (click here). GH exerts its anabolic effects in skeletal tissue, muscle, and adipose tissues(1).
Table 1.0 Anabolic effects of growth hormone
| Tissue | Effects |
| Skeletal tissue | 1) Synthesis of type 1 collagen
2) IGF-1 mediated linear growth (stimulation of chondroblasts) |
| Adipose tissue | 1) Promotes lipolysis by potentiating the effects on hormone-sensitive lipase (see figure 1.10)
2) Glucose uptake |
| Skeletal muscle | Protein synthesis and muscle growth |
Adapted from Root AW, Root MJ (2002) Clinical pharmacology of human growth hormone and its secretagogues. Curr Drug Targets Immune Endocr Metab Disord 2:27–52
Adipocyte and growth hormone physiology
There are four variants of adipocytes: brown, white, pink, and beige. This classification system is based on their location and physiologic role. White adipocyte physiology and the importance of growth hormone in fat storage and mobilization will be reviewed in this section.
White adipose tissue (WAT) is primarily involved in the storage and mobilization of fatty acids. Two enzyme systems – Lipoprotein lipase (LPL) and hormone-sensitive lipase (HSL) play essential roles in these energy-status-dependent tasks.
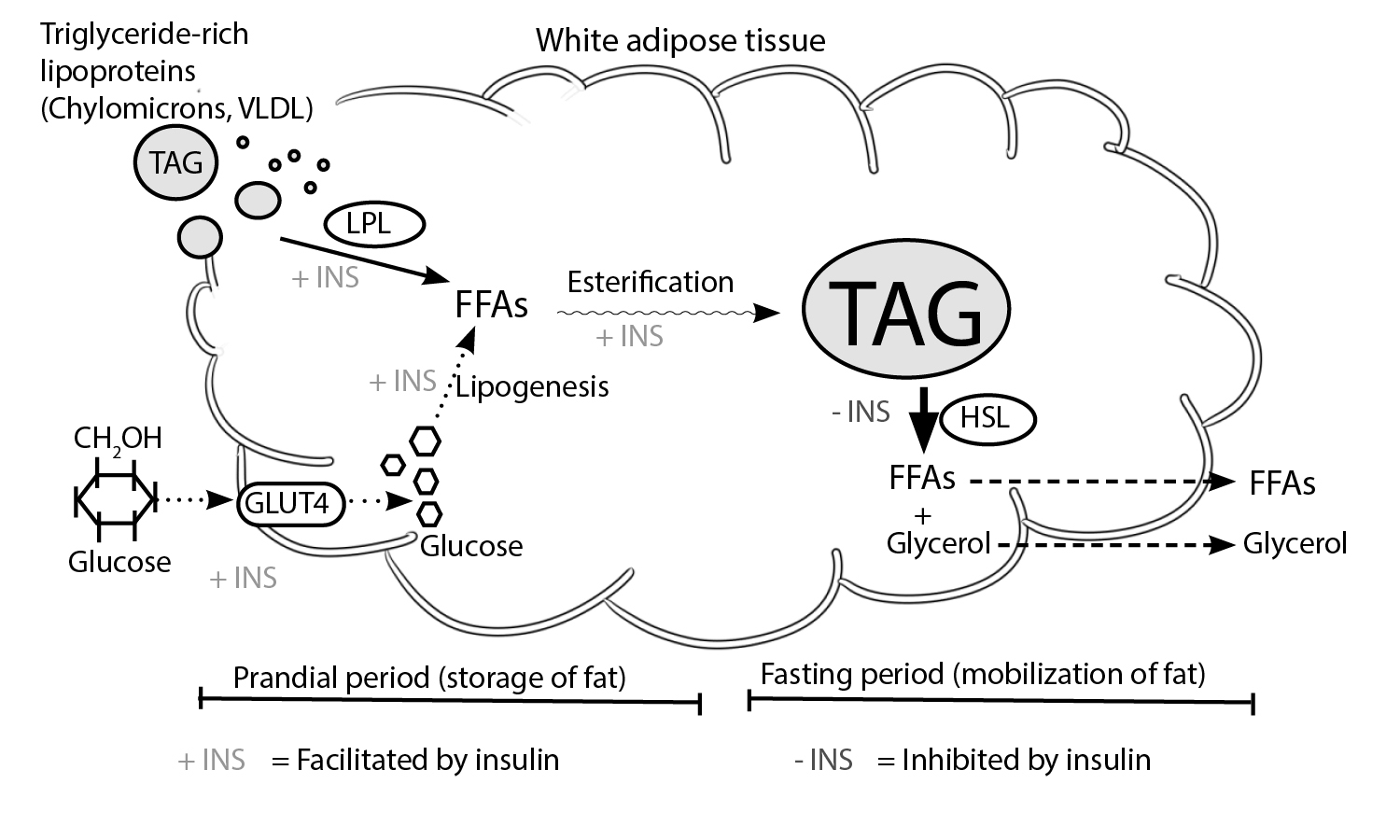
Fig. 1.0 The regulation of fat storage and mobilization by growth hormone. Storage of fat occurs in the prandial period, a process that involves the hydrolysis of triglycerides (TAG) in triglyceride-laden lipoproteins into free fatty acids (FFAs). LPL-induced storage of FFAs is facilitated by insulin(2) and inhibited by growth hormone(3). Also, glucose is transferred from the circulation into WAT by glucose transporter 4 (GLUT4) after a meal. The conversion of glucose into FFAs is also facilitated by insulin (4). Ultimately, FFAs are esterified into TAGs. During periods of energy deficit, hormone-sensitive lipase (HSL) mediates the conversion of stored repositories of TAG into FFAs and glycerol. These, in turn, become substrates for gluconeogenesis. Growth hormone facilitates HSL-induced lipolysis of TAG into FFAs and glycerol(5). Conversely, insulin inhibits this fat mobilization step. + = shows stimulatory factors , – = shows inhibitory factors. Redrawn and modified from Frayn et al. (1995) Coordinated regulation of hormone-sensitive lipase and lipoprotein lipase in human adipose tissue in vivo: Implications for the control of fat storage and fat mobilization. Adv Enzyme Regul 35:163–178)
Growth plate and growth hormone physiology
The growth plate (physis) is an anatomically distinct region composed of cartilage and with three physiologic zones critical in longitudinal bone growth (hypertrophic, proliferative, and resting zones)(6). Growth hormone promotes the proliferation of chondrocytes in the resting zone. IGF-1, produced by local chondrocytes, under trophic stimulation from GH, promotes the expansion of chondrocytes in all three zones of the growth plate(6,7).
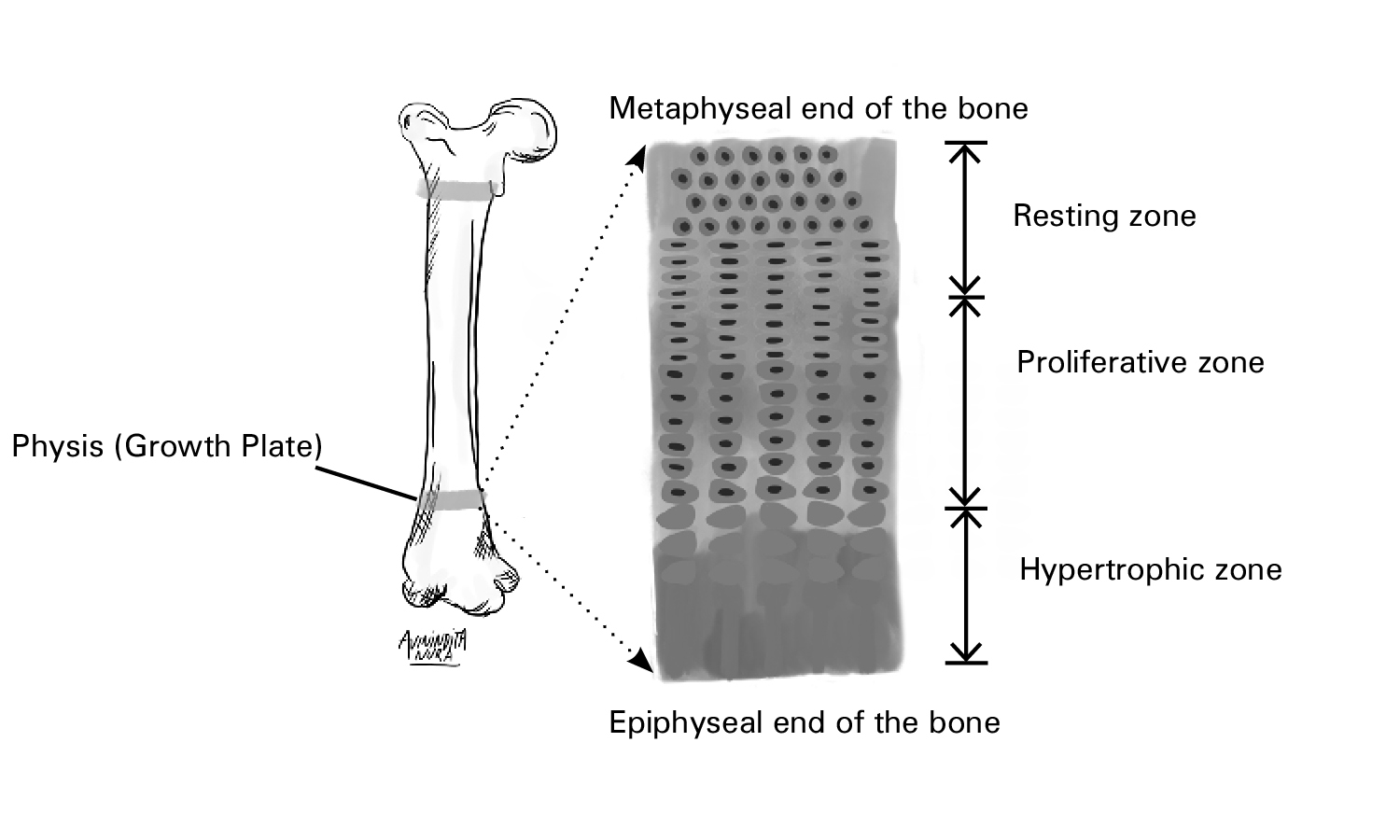
Fig. 1.1 The configuration of the three physiologic zones of the human growth plate. Growth plates are present at the ends of tubular bones. There is a sequential arrangement of the hypertrophic, proliferative, and resting zones from the epiphyseal to the metaphyseal ends of the bone(6). Nilsson O et al. (2005) Endocrine regulation of the growth plate. Horm Res 64:157–165
Mechanism of action
Recombinant human GH(rhGH) with an amino acid sequence similar to endogenous 22-kiloDalton(KDa) growth hormone(8,9) binds to the GH receptor and promotes the downstream effects of endogenous GH(10).
Practice Guide
- Common side effects of GH replacement in adults include myalgia, arthralgia, peripheral edema, and carpal tunnel syndrome. De-escalation in the dose of GH improves and, in some cases, leads to the amelioration of these symptoms(11). Prepubertal breast development, slipped capital femoral epiphysis, and benign intracranial hypertension can occur in children on GH therapy(12).
- The beneficial effects of growth hormone therapy in patients with AGHD include an improvement in lipid profiles, diastolic blood pressure, insulin sensitivity, and lean body mass to fat body mass ratio(13).
Clinical Trial Evidence
There are few high-quality, large placebo-controlled randomized clinical trials (RCTs) assessing the safety and efficacy of growth hormone replacement therapy(14).
In this randomized controlled trial, 43 subjects with AGHD in the setting of hypopituitarism were randomized to either placebo or recombinant human GH. The primary outcome included various surrogate cardiovascular endpoints, including visceral adiposity, insulin resistance, C-reactive protein (CRP), total cholesterol, and high-density lipoprotein. There was a reduction in high-sensitivity CRP by 38.2 vs 18.2 (p-value = 0.03), comparing GH to placebo. There was a statistically significant increase in IGF-1, comparing GH to placebo. Other demonstrable benefits of GH replacement included a decrease in total cholesterol and an increase in high-density lipoprotein(15).
References
- Root AW, Root MJ. Clinical pharmacology of human growth hormone and its secretagogues. Curr Drug Targets Immune Endocr Metab Disord. 2002 Apr;2(1):27–52.
- Frayn KN, Coppack SW, Fielding BA, Humphreys SM. Coordinated regulation of hormone-sensitive lipase and lipoprotein lipase in human adipose tissue in vivo: Implications for the control of fat storage and fat mobilization. Adv Enzyme Regul. 1995 Jan 1;35:163–78.
- Hjelholt AJ, Søndergaard E, Pedersen SB, Møller N, Jessen N, Jørgensen JOL. Growth hormone upregulates ANGPTL4 mRNA and suppresses lipoprotein lipase via fatty acids: Randomized experiments in human individuals. Metabolism. 2020;105:154188.
- Song Z, Xiaoli AM, Yang F. Regulation and Metabolic Significance of De Novo Lipogenesis in Adipose Tissues. Nutrients [Internet]. 2018 Sep 29 [cited 2020 Jun 20];10(10). Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6213738/
- Kopchick JJ, Berryman DE, Puri V, Lee KY, Jorgensen JOL. The effects of growth hormone on adipose tissue: old observations, new mechanisms. Nat Rev Endocrinol. 2020 Mar;16(3):135–46.
- Nilsson O, Marino R, De Luca F, Phillip M, Baron J. Endocrine regulation of the growth plate. Horm Res. 2005;64(4):157–65.
- Shim KS. Pubertal growth and epiphyseal fusion. Ann Pediatr Endocrinol Metab. 2015 Mar;20(1):8–12.
- Wallace JD, Cuneo RC, Bidlingmaier M, Lundberg PA, Carlsson L, Boguszewski CL, et al. Changes in Non-22-Kilodalton (kDa) Isoforms of Growth Hormone (GH) after Administration of 22-kDa Recombinant Human GH in Trained Adult Males. J Clin Endocrinol Metab. 2001 Apr 1;86(4):1731–7.
- Saugy M, Robinson N, Saudan C, Baume N, Avois L, Mangin P. Human growth hormone doping in sport. Br J Sports Med. 2006 Jul;40(Suppl 1):i35–9.
- Rezaei M, Zarkesh-Esfahani SH. Optimization of production of recombinant human growth hormone in Escherichia coli. J Res Med Sci Off J Isfahan Univ Med Sci. 2012 Jul;17(7):681–5.
- Díez JJ, Sangiao-Alvarellos S, Cordido F. Treatment with Growth Hormone for Adults with Growth Hormone Deficiency Syndrome: Benefits and Risks. Int J Mol Sci [Internet]. 2018 Mar 17 [cited 2020 Jun 4];19(3). Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5877754/
- Souza FM, Collett-Solberg PF. Adverse effects of growth hormone replacement therapy in children. Arq Bras Endocrinol Metabol. 2011 Nov;55(8):559–65.
- Maison P, Griffin S, Nicoue-Beglah M, Haddad N, Balkau B, Chanson P. Impact of Growth Hormone (GH) Treatment on Cardiovascular Risk Factors in GH-Deficient Adults: A Metaanalysis of Blinded, Randomized, Placebo-Controlled Trials. J Clin Endocrinol Metab. 2004 May 1;89(5):2192–9.
- Ramos-Leví AM, Marazuela M. Treatment of adult growth hormone deficiency with human recombinant growth hormone: an update on current evidence and critical review of advantages and pitfalls. Endocrine. 2018;60(2):203–18.
- Beauregard C, Utz AL, Schaub AE, Nachtigall L, Biller BMK, Miller KK, et al. Growth Hormone Decreases Visceral Fat and Improves Cardiovascular Risk Markers in Women with Hypopituitarism: A Randomized, Placebo-Controlled Study. J Clin Endocrinol Metab. 2008 Jun;93(6):2063–71.
Kindly Let Us Know If This Was helpful? Thank You!



