Introduction
Osteoporosis is a condition in which the bones become weak and brittle, making them more susceptible to fractures. While osteoporosis can affect people of any age, it is most commonly seen in older adults.
There are a number of different treatments available for osteoporosis, and the best course of treatment will vary depending on the individual. In general, however, medical treatment for osteoporosis focuses on two main goals: increasing bone density and reducing the risk of fractures.
A variety of medications can be used to achieve these goals, including bisphosphonates, calcitonin, PTH analogs, PTHrp analogs, calcitonin, SERMs, and romosozumab. Their mechanism of action will be briefly reviewed next.
Bisphosphonates
Background
Nitrogen-containing bisphosphonates (N-BPs) are a group of drugs that are commonly used to treat osteoporosis. They work by inhibiting the activity of osteoclasts, cells that break down bone tissue. N-BPs were first synthesized in the 1950s, but it was not until the 1970s that they were shown to have potential therapeutic use. The first N-BP to be approved for medical use was etidronate, which was approved in 1977 to treat paget’s disease, a condition characterized by bone pain and deformity. Since then, several different N-BPs have been developed, each with its unique advantages and disadvantages. Today, N-BPs are an essential part of the treatment arsenal for osteoporosis, and their discovery has had a profound impact on the lives of millions of people worldwide.
Mechanism of action
The farnesyl pyrophosphate synthase (FPPS) protein plays a critical role in the mevalonic acid-cholesterol synthesis pathway in various tissues, including osteoclasts. While the farnesyl pyrophosphate molecule is required for the construction of new cholesterol molecules, it is also necessary for the proper maintenance of cellular membranes.
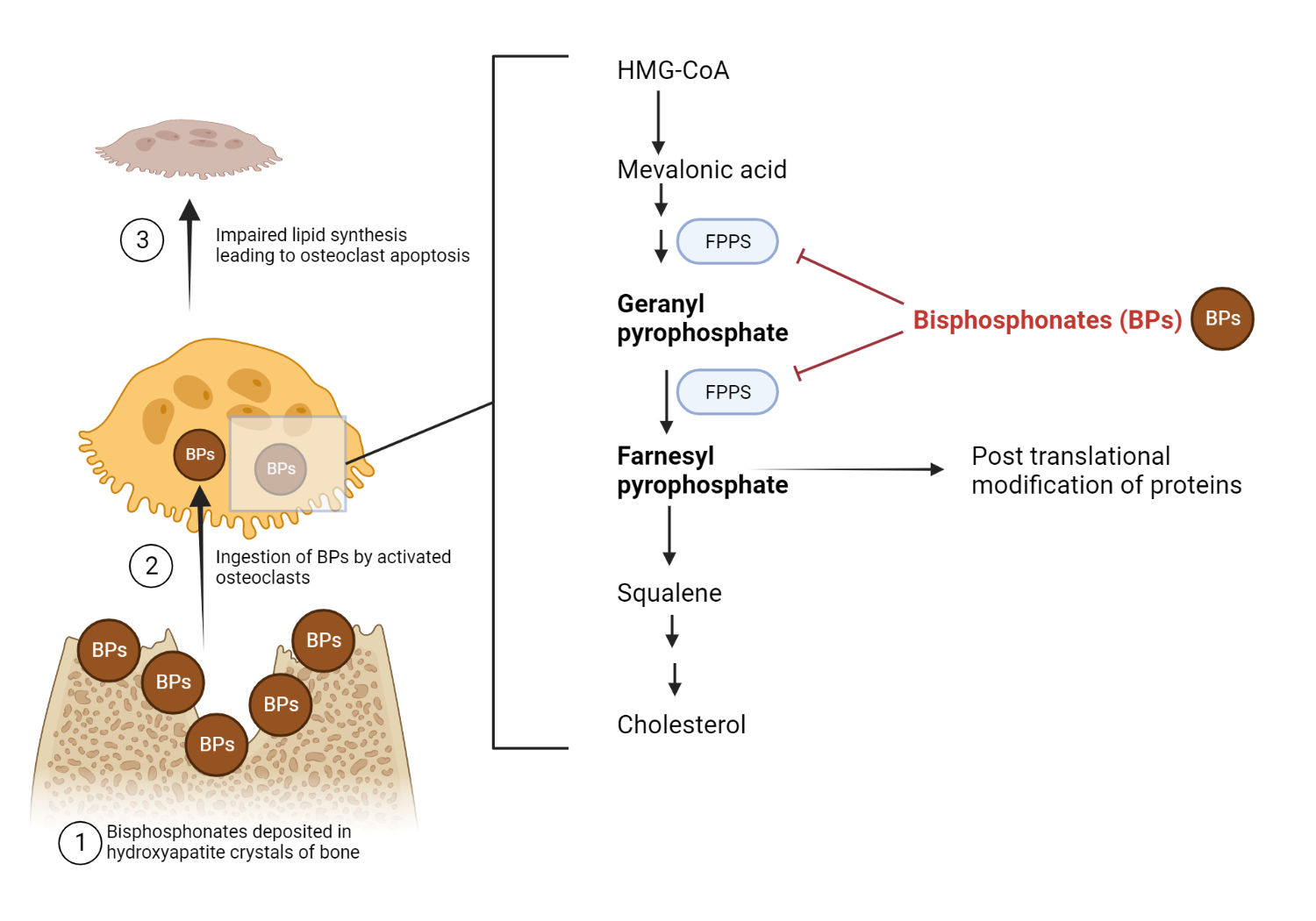
Nitrogen-containing bisphosphonates are a type of drug that inhibits the enzyme farnesyl pyrophosphate synthase (FPPS). Although farnesyl pyrophosphate synthase is available in various tissues, its inhibition by bisphosphonates occurs mainly in bone. This effect is thought to be due to ability of bisphosphonate’s to bind almost exclusively to bone minerals present in hydroxyapatite crystals.
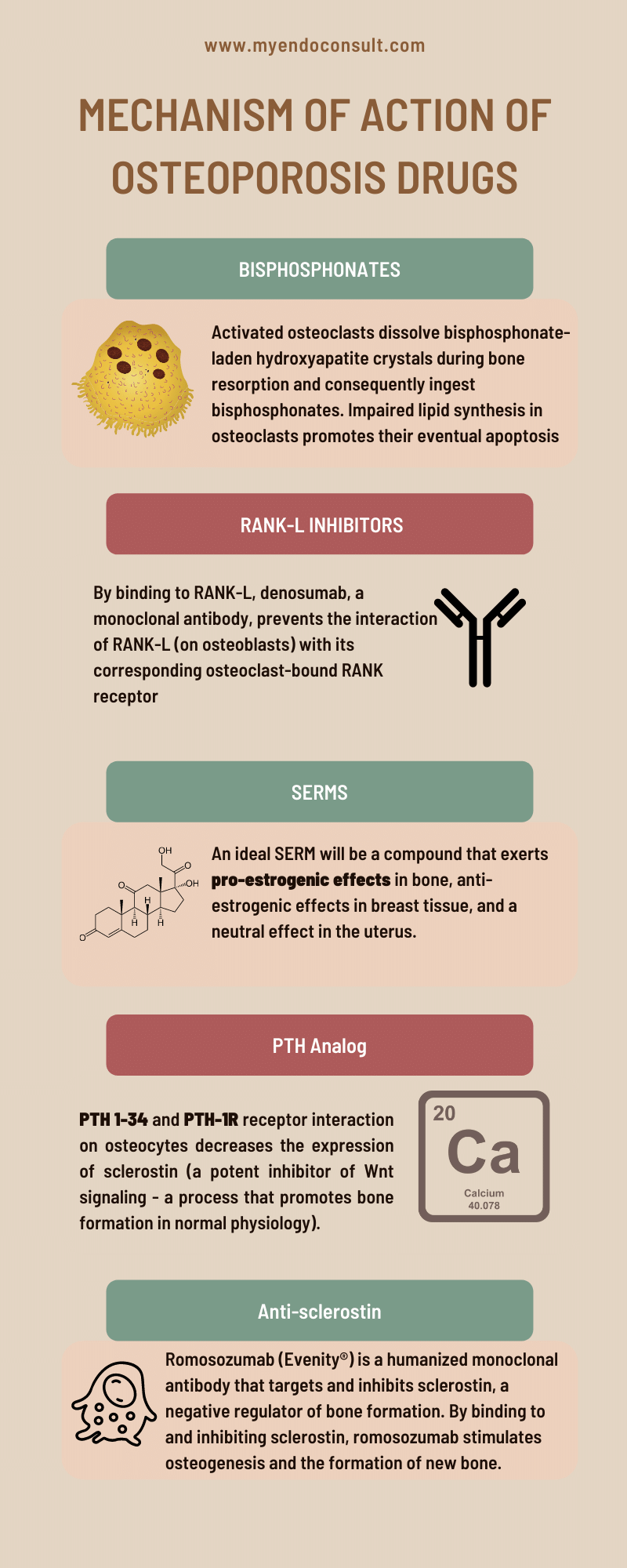
When osteoclasts resorb bone, they dissolve the mineral component, hydroxyapatite, composed of calcium and phosphate. In patients who have been treated with bisphosphonates, these drugs become incorporated into the hydroxyapatite crystals. The bisphosphonate is released and ingested as the osteoclast dissolves the hydroxyapatite crystal. Once inside the cell, the bisphosphonate inhibits farnesyl pyrophosphate synthase, an enzyme involved in cholesterol synthesis. This action results in apoptosis, or cell death, of the osteoclast. Consequently, by inhibiting FPPS in osteoclasts, bisphosphonates can help prevent bone loss and improve bone mineral density.
RANK-L modulators (Denosumab)
Background
The development of denosumab began with the identification of the RANKL protein in 1999. In 2003, early animal studies showed that denosumab was effective in preventing bone loss. The first clinical trials in humans were conducted in 2004, and the drug was approved by the US Food and Drug Administration (FDA) in 2010. Since then, denosumab has become an important treatment option for patients with osteoporosis and other bone-related conditions.
Mechanism of action
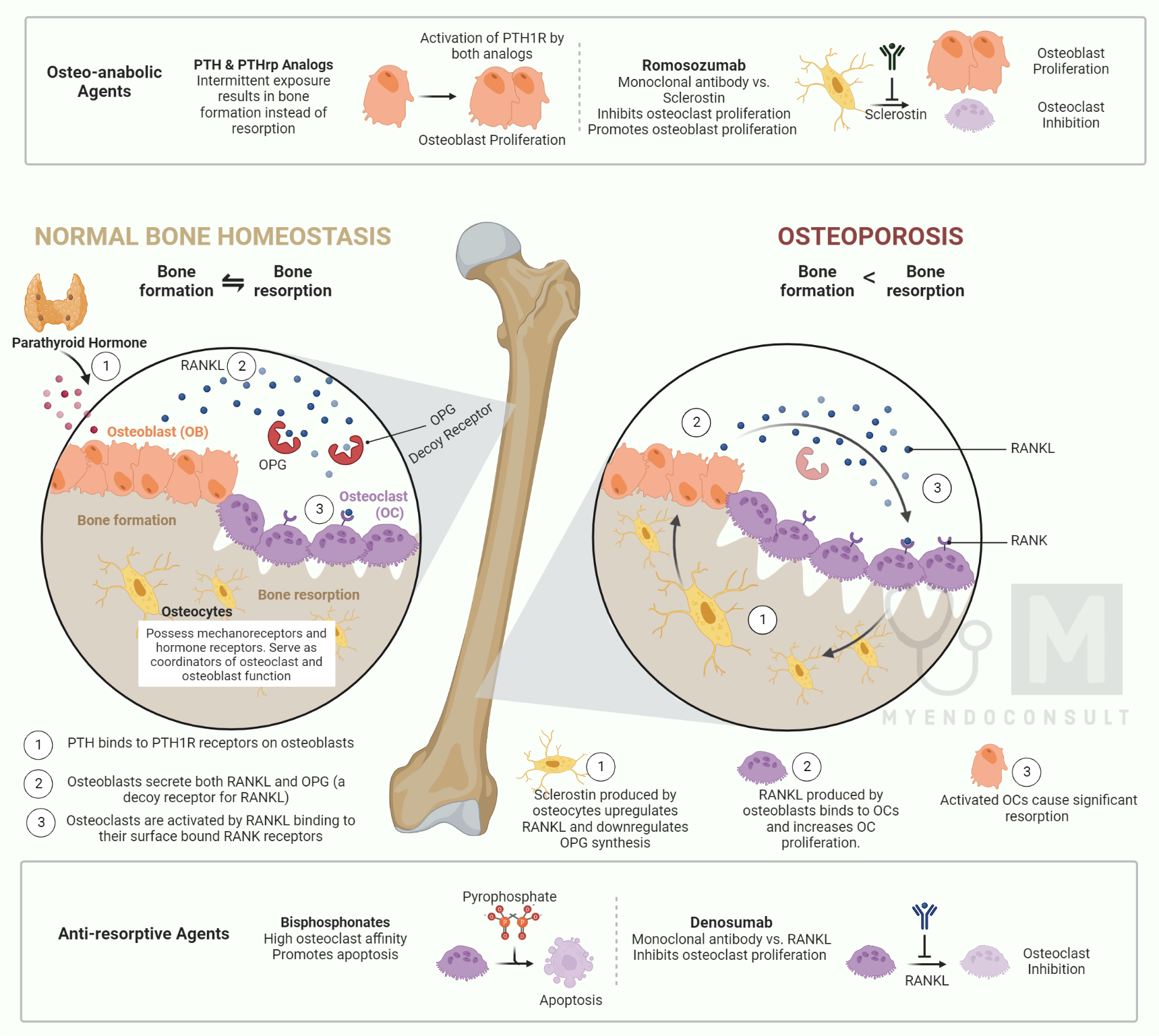
Osteoclastogenesis is the process by which osteoclasts are formed. It is essential for bone remodeling, homeostasis, and normal healing after a fracture.
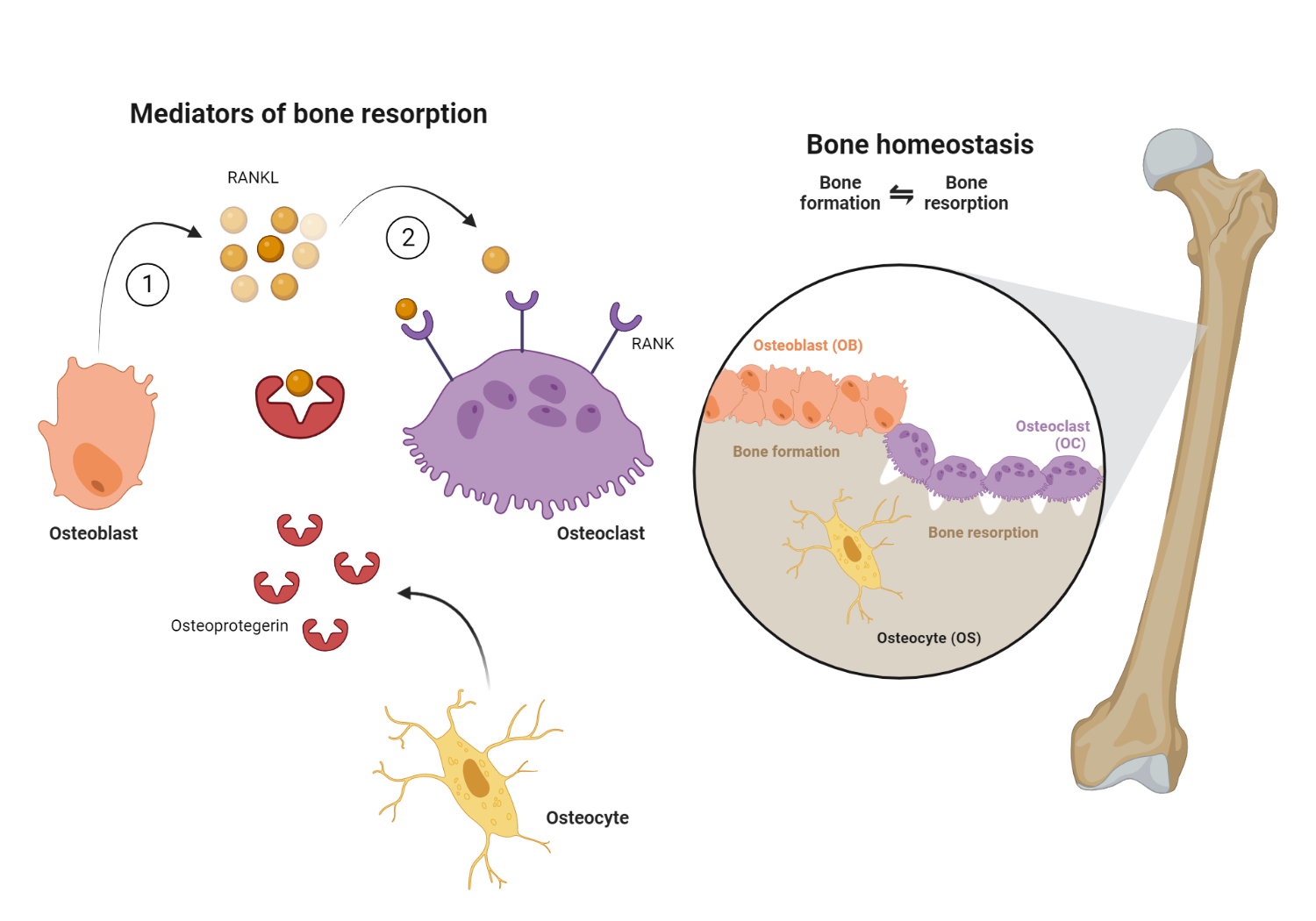
The formation of osteoclasts requires the interaction of two molecules: RANK (receptor activator of nuclear factor kappa-B) and RANK-L (receptor activator of nuclear factor kappa-B ligand). RANK is expressed on the surface of osteoclast precursors, while RANK-L is expressed on the surface of stromal cells and osteoblasts. When RANK-L binds to RANK, it triggers a series of events that leads to the activation of osteoclasts. This process is regulated by a number of other molecules, including cytokines and transcription factors. Understanding the mechanism of RANK and RANK-L interaction is essential for developing drugs that can target this process and prevent or treat disorders associated with bone loss. Denosumab is a monoclonal antibody that binds to RANK-L, thereby preventing the interaction of RANK-L present on osteoblasts with its complementary osteoclast-bound RANK receptor. This results in the inhibition of osteoclastogenesis, and subsequent bone resorption.
Selective Estrogen Receptor Modulators (SERMs)
Background
SERMs are a class of compounds that act on traditional estrogen receptors in a tissue-specific manner. Depending on the tissue, they can act as agonists or antagonists on the receptors. This means that they can either stimulate or inhibit estrogenic activity, depending on the tissue in which they bind. For example, the ideal SERM would have high binding affinity for the ER, agonist activity in bone, and antagonist activity in breast and endometrium.
Mechanism of action
17β-estradiol, more commonly known as estrogen, is a hormone that is produced by the ovaries. Estrogen plays an important role in regulating the menstrual cycle and is also involved in the development of female reproductive organs and secondary sex characteristics. In addition to its reproductive functions, estrogen also plays a role in bone health. Studies have shown that estrogen inhibits bone resorption, which is the process by which osteoclasts break down bone tissue. This effect is mediated by estrogen’s ability to bind to the estrogen receptor alpha (ERα) in osteoblasts and upregulate the expression of osteoprotegerin (OPG), a protein that acts as a decoy receptor for RANK-L, a protein required for osteoclastogenesis. By binding to RANK-L and preventing it from interacting with osteoblasts, OPG inhibits bone resorption. In addition to its effects on OPG expression, estrogen also decreases the ability of osteoclasts to create an acidic environment necessary for bone resorption. These effects work together to protect bones from deterioration and prevent bone loss.
PTH Analogs
Background
PTH analogs are a class of drugs that mimic the action of parathyroid hormone (PTH), a hormone that helps to regulate calcium metabolism in the body. The peptide was originally designed as a PTH analog that would have bone anabolic activity without inducing hypercalcemia (high blood calcium levels). Lilly subsequently applied for patents on the use of teriparatide for the treatment of osteoporosis and several other indications.
Mechanism of action
Parathyroid hormone (PTH) is a peptide hormone secreted by the parathyroid gland that regulates calcium homeostasis. PTH acts on bone, kidney, and intestine to regulate calcium and phosphate metabolism. In bone, PTH promotes resorption of calcium and release of phosphate into the circulation, which raises serum calcium levels and leads to feedback inhibition of PTH secretion. PTH also stimulates osteoblast activity and promotes new bone formation. Thus, PTH serves a dual role in bone metabolism by promoting both bone resorption and formation. The net effect of PTH on bone is determined by the balance between its stimulatory effect on bone formation and its inhibitory effect on bone resorption. Teriparatide is a synthetic peptide that mimics the effects of parathyroid hormone. By binding to the PTH receptor, it increases bone formation and stimulates osteoblasts.
PTHrp analogs
Background
Tymlos is a synthetic hormone that is used to treat postmenopausal osteoporosis. It is an analog of parathyroid hormone (PTH) and works by stimulating new bone formation and inhibiting bone resorption. Tymlos was approved by the US Food and Drug Administration (FDA) in April 2017. It is manufactured by Radius Health, Inc. The FDA review process for Tymlos included two randomized, double-blind, placebo-controlled clinical trials involving over 3,700 women with postmenopausal osteoporosis. The trials showed that treatment with Tymlos increased bone density and reduced the incidence of new fractures compared to placebo
Mechanism of action
PTHrp is a protein that is secreted by the placenta, growth plate, and mammary tissue. PTHrp shares some structural similarities with PTH, which explains why PTHrp serves as a ligand for the PTH-1R receptor. Abaloparatide (tymlos®) is a man-made hormone that is similar to a naturally occurring hormone called parathyroid hormone-related protein (PTHrP) binds to the PTH-1R receptor and stimulates bone formation.
Anti-sclerostin modulators
Background
On September 25, 2019, the U.S. Food and Drug Administration (FDA) approved Evenity™ (romosozumab-aqqg) for the treatment of postmenopausal women with osteoporosis at high risk for fracture. Evenity is a monoclonal antibody that targets and inhibits sclerostin, a protein that inhibits bone formation.
Mechanism of action
Sclerostin (encoded by the SOST gene) is secreted by mature osteocytes embedded in bone. Sclerostin plays a dual role in bone metabolism by negatively regulating osteoblast differentiation (inhibition of bone formation) while promoting osteoclast formation (promotion of bone resorption). By binding to LRP5/6, sclerostin inhibits the Wnt/β-catenin signaling pathway, leading to impaired bone formation. Furthermore, sclerostin upregulates RANK-L and downregulates OPG synthesis, which increases bone resorption.
Mechanism of action
Romosozumab (Evenity®) is a humanized monoclonal antibody to endogenous sclerostin, which lifts the inhibition of the Wnt/β-catenin signaling pathway by sclerostin.
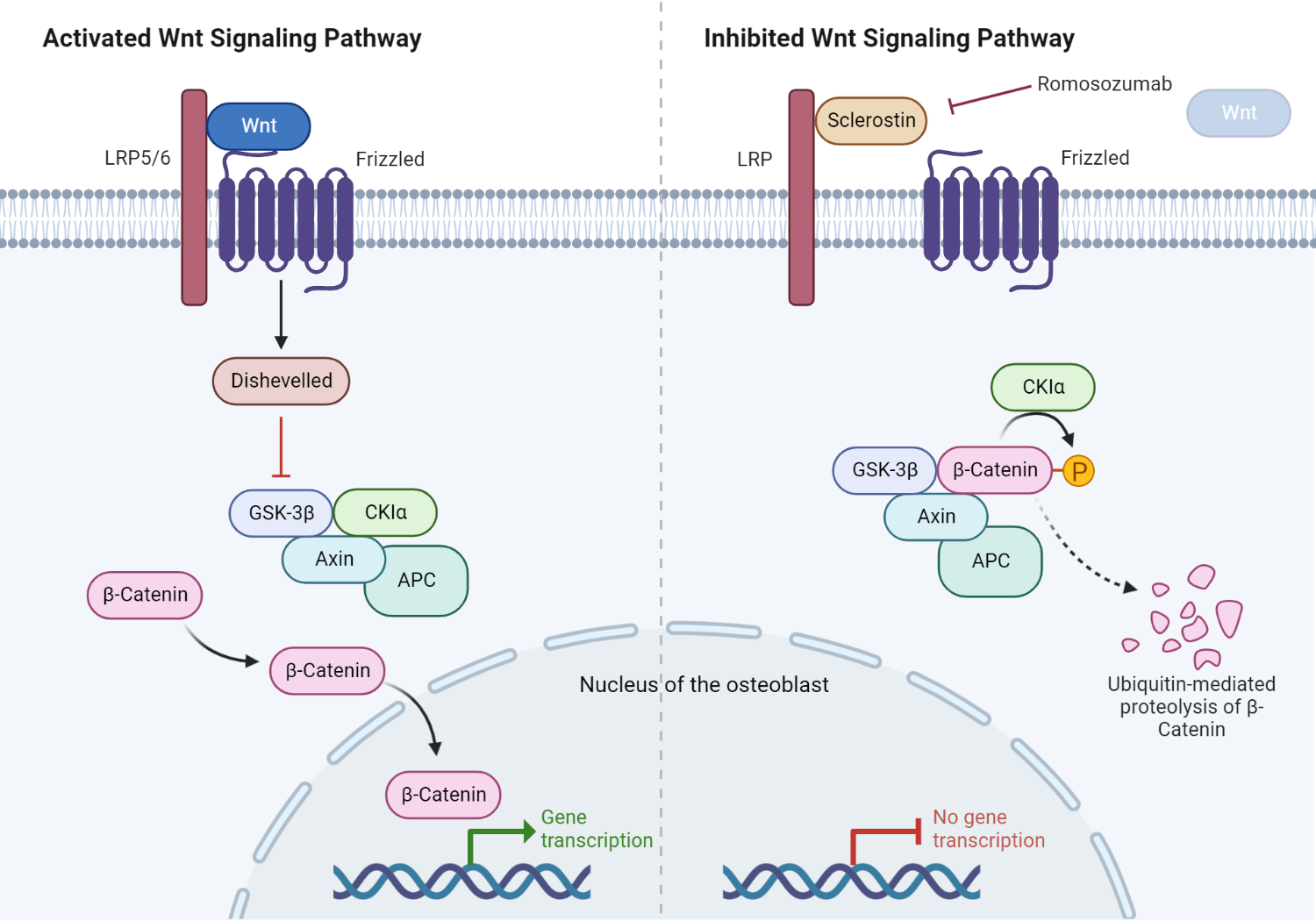
Role of sclerostin in the Wnt/β-catenin signaling pathway.
In normal physiology, the Wnt ligand binds to its cognate receptor, the LRP5/6 complex, on the plasma membrane of osteoblasts. The activated LRP5/6 complex subsequently activates an intracytosolic protein called Disheveled (DSH). Consequently, DSH turns off glycogen synthase kinase 3β (GSK-3β), an inactivating system for β-catenin. This ultimately allows β-catenin to escape proteosomal degradation in the cytosol of the osteoblast. β-catenin then enters the nucleus, promoting the transcription and translation of Wnt target genes critical in osteoblast differentiation and activation. Furthermore, sclerostin occupies the Wnt binding site on LRP5/6, inhibiting downstream processes critical in protecting β-catenin. The presence of sclerostin, therefore, inhibits bone formation. Romosozumab promotes bone formation by inhibiting sclerostin.
Sclerostin is a protein that is produced by osteocytes, which are cells critical in regulating the function of osteoblasts and osteocytes. Sclerostin inhibits the activity of osteoblasts, which are cells that contribute to the formation of new bone. In addition, sclerostin promotes the activity of osteoclasts, which are cells that break down old bone. As a result, sclerostin plays an important role in regulating the rate of bone formation and resorption. Romosozumab (Evenity®) is a humanized monoclonal antibody that targets and inhibits sclerostin, a negative regulator of bone formation. By binding to and inhibiting sclerostin, romosozumab stimulates osteogenesis and the formation of new bone. Additionally, romosozumab increases bone mass by enhancing bone turnover, resulting in increased levels of both osteoblasts and osteoclasts. The net effect is an increase in bone density and strength.
Calcitonin
Background
Nasal calcitonin (NCT) is used to treat osteoporosis and other bone diseases. It is a synthetic form of the hormone calcitonin, which is found naturally in the body. NCT works by inhibiting bone reabsorption, preventing calcium loss and promoting bone formation. The discovery of NCT began in the early 1960s, when researchers noticed that calcitonin injections could reduce bone loss in rats. This led to clinical trials in humans, which showed that NCT was effective in treating osteoporosis. Today, NCT is available as a nasal spray or injectable, and it is an important treatment for preserving bone health.
Mechanism of action
Calcitonin is a hormone that inhibits osteoclast activity, making it a suitable agent for treating osteoporosis. When osteoclasts are active, they break down bone and release calcium into the bloodstream. This can lead to a decrease in bone density and an increased risk of fractures. Calcitonin helps to prevent this by inhibiting the activity of osteoclasts.
References
- Drake MT, Clarke BL, Khosla S (2008) Bisphosphonates: mechanism of action and role in clinical practice. Mayo Clin Proc 83:1032–1045
- Roelofs AJ, Stewart CA, Sun S, et al (2012) Influence of bone affinity on the skeletal distribution of fluorescently labeled bisphosphonates in vivo. J Bone Miner Res 27:835–847
- Zaheer S, LeBoff M, Lewiecki EM (2015) Denosumab for the Treatment of Osteoporosis. Expert Opin Drug Metab Toxicol 11:461–470
- Tam CS, Heersche JNM, Murray TM, Parsons JA (1982) Parathyroid Hormone Stimulates the Bone Apposition Rate Independently of Its Resorptive Action: Differential Effects of Intermittent and Continuous Administration. Endocrinology 110:506–512
- Estell EG, Rosen CJ (2021) Emerging insights into the comparative effectiveness of anabolic therapies for osteoporosis. Nat Rev Endocrinol 17:31–46
- Padhi D, Allison M, Kivitz AJ, Gutierrez MJ, Stouch B, Wang C, Jang G (2014) Multiple doses of sclerostin antibody romosozumab in healthy men and postmenopausal women with low bone mass: a randomized, double-blind, placebo-controlled study. J Clin Pharmacol 54:168–178
- Yang D, Singh R, Divieti P, Guo J, Bouxsein M, Bringhurst FR (2007) Contributions of Parathyroid Hormone (PTH)/PTH-Related Peptide Receptor Signaling Pathways to the Anabolic Effect of PTH on Bone. Bone 40:1453–1461
Images(s) Courtesy
MyEndoConsult
Kindly Let Us Know If This Was helpful? Thank You!


