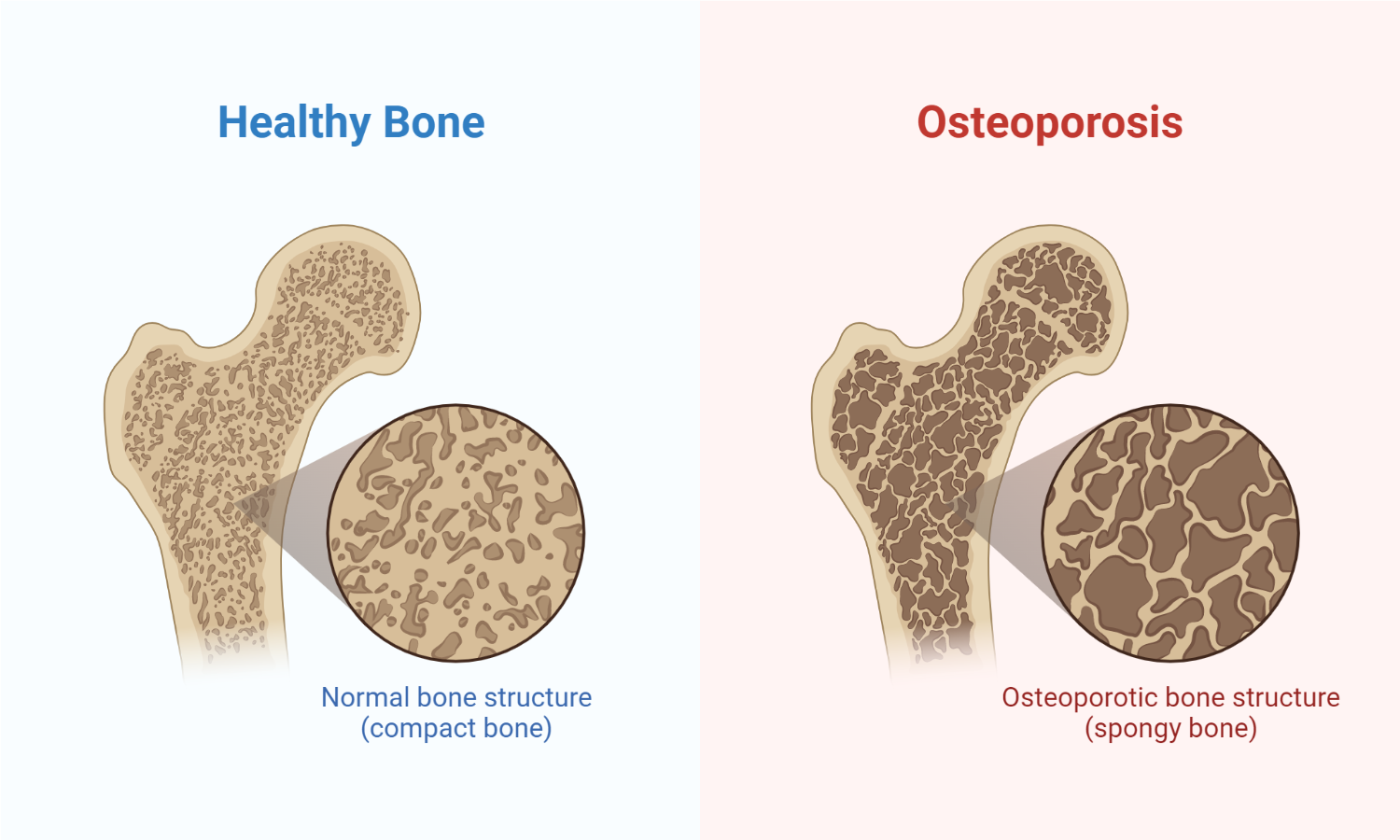
Osteoporosis and osteomalacia are known causes of metabolic bone disease. Although there is a significant difference both conditions, they share certain common features. These include a reduction in bone mineral density (BMD) on bone densitometry (DXA), fractures and bone pain. Indeed, in most cases, telling the difference between osteoporosis and osteomalacia is difficult in the absence of a bone biopsy.
This is because histologically, osteomalacia is characterized by a mineralization defect of bone while osteoporosis typically has normal bone composition (but reduced bone volume).
Difference between osteoporosis and osteomalacia
| Clinical feature | Osteoporosis | Osteomalacia |
| 25-OH vitamin D | Normal, low or high vitamin D | Usually low |
| Vertebral fractures | May be present | Usually absent |
| Volume of osteoid | Normal or Decreased | Increased |
| Symptoms | Silent disease | Symptomatic (muscle pain, muscle weakness, increased fall risk and bone pain) |
| Biochemical picture | Usually normal calcium, phosphate and bone-specific alkaline phosphatase | Associated with hypocalcemia, hypophosphatemia and a high alkaline phosphatase. |
Osteomalacia/Osteoporosis discriminant index
| Parameter | Score |
| Limb pain | 1 |
| Proximal myopathy | 1 |
| [Ca x P] 1.62-2.41 | 1 |
| [Ca x P] 1.21-1.61 | 2 |
| [Ca x P] <1.21 | 3 |
| ALP 75-100 | 1 |
| ALP 101-201 | 2 |
| ALP 201-300 | 3 |
| ALP >300 | 4 |
| 25(OH)D 5-12.5 | 1 |
| 25(OH)D <5 | 2 |
| Presence of pseudofracture | 1 |
[Ca x P] Calcium-phosphate product in mmol/L
ALP Alkaline phosphatase
25(OH)D 25 hydroxyvitamin D in nmol/L
Interpretation of the discriminant index
% total index score = [total score obtained / total possible score] x 100
>35% = osteomalacia
15-35% = bone biopsy indicated
<15% = Unlikely to have osteomalacia. High probability of osteoporosis if there is a vertebral fracture.
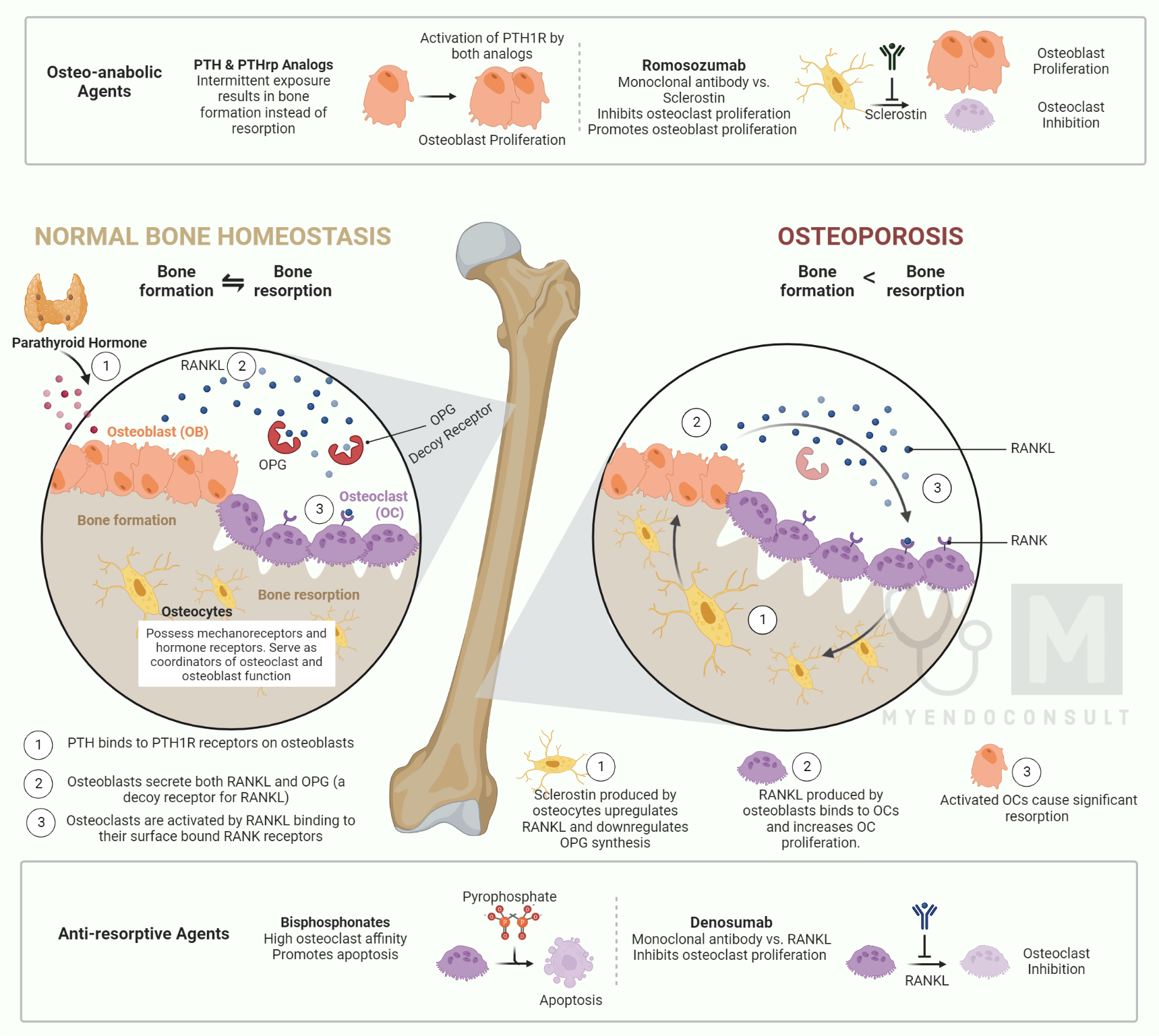
What is osteoporosis?
Osteoporosis is the physiological loss of bone minerals with advancing age. Osteoporosis is characterized by a morphometrically measurable decrease in the volumetric bone density of the spongiosa, a reduction of the thickness of the cortical bone, and an enlargement of the Haversian compartments of bone.
What is osteomalacia?
Osteomalacia is characterized by an increase in unmineralized osteoid due to a defect in the mineralization of the bone matrix. Amorphous calcium phosphate is deposited into the osteoid, which is only slowly converted into hydroxyapatite.
The pathognomonic finding is pseudo fractures/Milkman’s line/Looser’s zones noted on plain radiographs. Pseudofractures on plain xrays appear as horizontal radiolucent lines perpendicular to the imaged bone’s long axis (right angles to the involved cortex). It characteristically does not traverse the entire cortex of the involved bone. The most common site for this condition includes the scapula, ribs, bilateral pubic rabi, and proximal femur. Interestingly these fractures tend to be bilaterally symmetrical.
 Xray of the proximal femur showing a pseudofracture.
Xray of the proximal femur showing a pseudofracture.
Vitamin D deficiency due to lack of nutritional intake, gastrointestinal malabsorption, genetic conditions, etc., are known causes of osteomalacia.
An increase in osteoid mass is associated with vitamin D deficiency; however, an increase in osteoid mass alone is no proof of a vitamin D deficiency. Osteomalacia connected with the widening of the osteoid seams also occurs in patients with a phosphate-deficient diet or with phosphate loss through the kidneys.
Osteoblasts and tetracycline deposits in osteoid seams are indications that bone formation is normal. Their absence suggests a mineralization defect such as that occurs in vitamin-D deficiency-related osteomalacia.
Tetracycline labeling of bone biopsy (normal bone). Tetracycline is typically administered on two separate occasions, separated by a few days. A well-formed double tetracycline label suggests normal bone mineralization. The main bone minerals are calcium and phosphate. They are present in the form of amorphous calcium phosphate and hydroxyapatite. Tetracycline is deposited in the transitional zones between osteoid and hydroxyapatite. The area of the surface marked with tetracycline gives an indication of the rate of the mineralization of the skeleton and, thus, of the formation of new bone
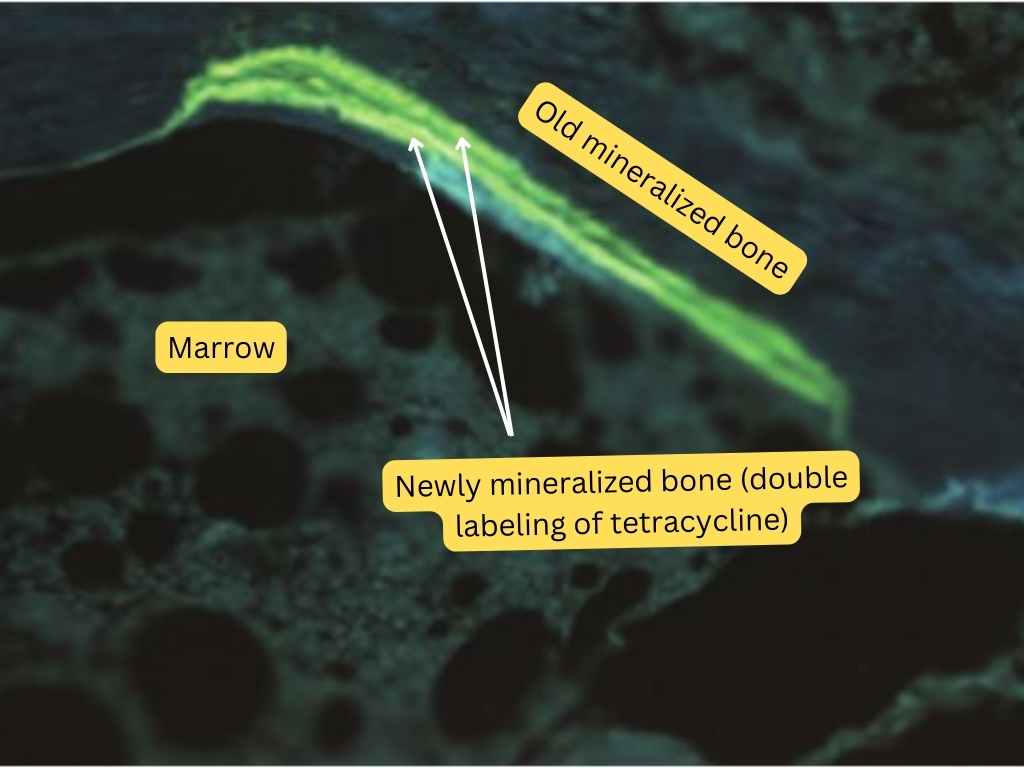 Tetracycline labeling on bone biopsy (normal bone)
Tetracycline labeling on bone biopsy (normal bone)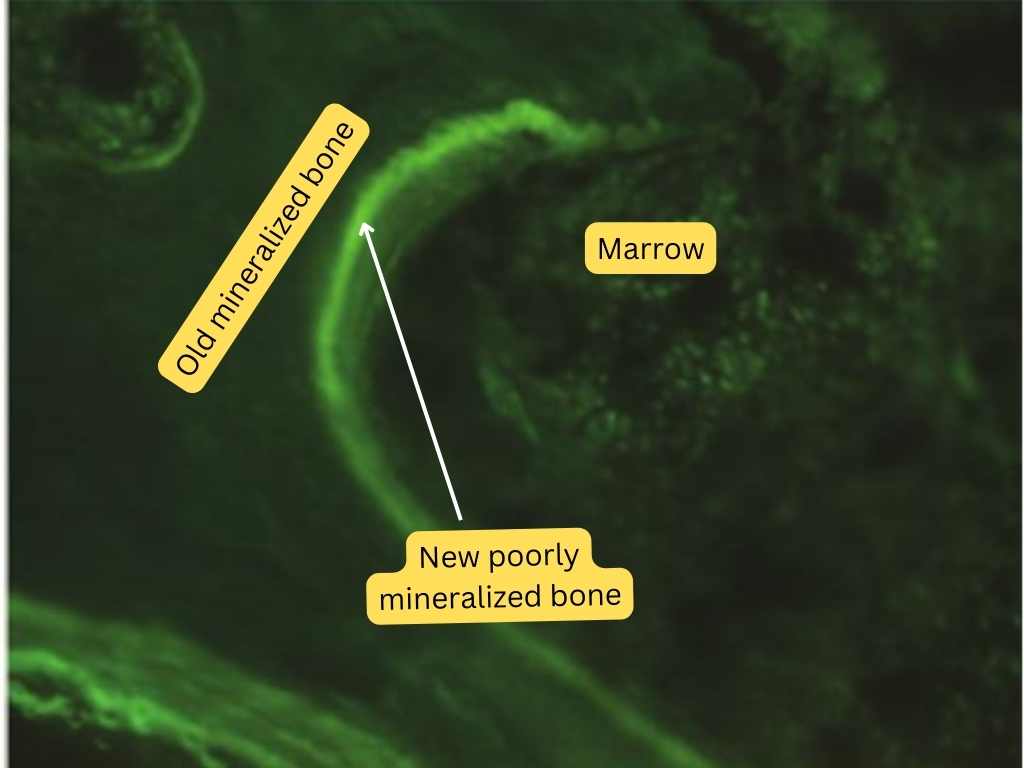 Tetracycline labeling on bone biopsy (osteomalacia). The absence of the classic double tetracycline label is suggestive of poor bone mineralization (e.g., osteomalacia)
Tetracycline labeling on bone biopsy (osteomalacia). The absence of the classic double tetracycline label is suggestive of poor bone mineralization (e.g., osteomalacia)
The pathogenesis of osteomalacia cannot be explained by the absence of a single factor, with the exception of vitamin D deficiency in infantile rickets and of nutritive vitamin D deficiency, which rarely occurs alone.
Osteomalacia develops in vitamin-D deficiency in children (rickets) or as a result of resistance to the action of vitamin D (renal insufficiency, vitamin D-resistant rickets). It also arises in steatorrhea after gastrectomy, in liver cirrhosis, with a phosphate-deficient diet, or a phosphate loss through the kidneys.
Biochemical features of vitamin D deficiency-related osteomalacia
Vitamin D deficiency is the dominant feature of infantile rickets. At first, the serum calcium concentration is usually only slightly reduced; the reduction later becomes severe, causing tetany. The serum phosphate concentration is low, and the phosphate excretion in the urine is elevated due to secondary hyperparathyroidism.
The alkaline phosphatase in the plasma is raised before and immediately after treatment with vitamin D and is a sign of increased osteoblastic activity. Parathyroid hormone deficiency alone, in contrast to vitamin D deficiency, does not lead to a massive increase in osteoid mass.
Malabsorptive causes of osteomalacia
In the malabsorption syndrome with steatorrhea, osteoporosis sometimes precedes osteomalacia and can be treated successfully with vitamin D.
Steatorrhea prevents normal intestinal absorption of the fat-soluble vitamin D, and furthermore, phosphate absorption is reduced since a significant proportion of the phosphate is delivered to the organism in the form of fats. Finally, protein absorption is decreased, which is of importance in the synthesis of the bone matrix.
Chronic kidney failure-related osteomalacia
In renal insufficiency, the reduction of the calcium concentration in the plasma is the predominant feature at first. The osteoid surface is generally increased, and tetracycline storage is limited to a few seams. The metabolic acidosis occurring in uremic patients probably also contributes to the inhibition of the mineralization of the skeleton. Bicarbonate leads to a reduction of calcium excretion in the stools and urine when serum calcium and phosphate are unaltered. The intestinal absorption of calcium is reduced even when concentrations of vitamin D in the plasma are normal. This is corrected only with pharmacological doses of vitamin D.
The phosphate concentration in the plasma is elevated due to the reduced glomerular filtration rate. Hyperphosphatemia also leads to hypocalcemia and sustains secondary hyperparathyroidism. The severity of secondary hyperparathyroidism can be reduced with phosphate binders or a phosphate-deficient diet.
During prolonged azotemia the osteomalacia disappears, but bone resorption increases, and the serum calcium is normalized during stimulation of the parathyroid hormone secretion.
Vitamin D-resistant rickets-associated osteomalacia
Vitamin D-resistant rickets result from a loss of phosphate through the kidneys. The calcium concentration in the plasma is normal, although calcium absorption from the gut and excretion through the kidneys is reduced. The state of phosphate loss is due primarily to a defect in the renal tubules.
Dr. Fuller Albright reported a case of vitamin D-resistant rickets in 1937. He postulated the underlying cause of rickets in his case report as being due to “an intrinsic resistance to the anti-rachitic action of vitamin D”.
The clinical features of rickets occur at a young age and classically manifest before the fusion of the growth plates. Rickets tends to affect the distal forearms, knees, and costochondral regions. The typical features include swelling of the costochondral joints (rachitic rosary), widening of the wrist joint, and an exaggerated genu varum in children.
Hereditary Vitamin D-Resistant Rickets Type 2 is an autosomal recessive condition due to a mutation in the vitamin D receptor gene, which results in impaired activity of vitamin D due to end-organ resistance to the action of 1,25-dihydroxy vitamin D.
Patients develop hypocalcemia with or without hypophosphatemia. There is another form of vitamin D-dependent rickets due to a mutation in the gene encoding the renal 1-alpha-hydroxylase enzyme.
Transformation of cartilage into bone involves a cycle of laying down of cartilage matrix, its resorption, and replacement by series of woven bone and then the eventual formation of mature lamellar bone.
Mineralization of newly formed osteoid is defective due to a lack of calcium and phosphorus, which results in the formation of defective bones unable to withstand mechanical stress. This leads to the typically exaggerated genu varum seen in rickets.
| Clinical feature | Underlying Cause |
| Decreased linear growth | Impaired mineralization in the growth plate |
| Tetany | Hypocalcemia |
| Hypotonia | Hypocalcemia |
| Recurrent respiratory infections | Hypotonia involving respiratory musculature results in an inability to clear airway secretions. |
How can HVDRR-II be differentiated from vitamin D resistance due to 1ɑ hydroxylase deficiency, biochemically?Low serum calcium and phosphate with high parathyroid hormone levels occur in both conditions. 1,25 hydroxyvitamin D3 is, however, low in 1ɑ hydroxylase deficiency but high in HVDRR.
Treatment with vitamin D in doses that would normally lead to vitamin D intoxication improves the mineralization of the skeleton. Calcium administration does not lead, as would be expected, to the improvement of osteomalacia. Calcium cannot be incorporated into the bones in the presence of a phosphate deficiency. Additional evidence that the rate of bone formation is slow comes from kinetic measurements, which show that the deposition rate is reduced and from a decrease of the alkaline phosphatase in the plasma and the excretion of hydroxyproline in the urine.
Even treatment with phosphate alone improves the absorption of calcium and phosphate in the gut and the mineralization to some extent. This is associated with a rise in the calcium deposition in osteoid. Furthermore, there is a decrease in the calcium concentration in the plasma and the calcium excretion in the urine and an increase in the alkaline phosphatase level in the plasma and in the urinary hydroxyproline excretion. All of these changes reflect secondary hyperparathyroidism.
Reference(s)
McKenna MJ, Freaney R, Casey OM, Towers RP, Muldowney FP. Osteomalacia and osteoporosis: evaluation of a diagnostic index. J Clin Pathol. 1983 Mar;36(3):245-52. doi: 10.1136/jcp.36.3.245. PMID: 6826778; PMCID: PMC498193.
Kindly Let Us Know If This Was helpful? Thank You!



