Introduction
Prolactinomas are the most commonly occurring hormone-secreting pituitary tumors, predominantly composed of monoclonal lactotroph cell adenomas. These tumors typically arise from sporadic mutations, but they also frequently occur in individuals with Multiple Endocrine Neoplasia type 1. Over 99% of prolactinomas are benign, with about 10% also co-secreting growth hormone due to a somatotroph or mammosomatotroph component.
Prolactin physiology
Prolactin, a hormone known for its diverse actions, plays significant roles in various bodily functions, often in conjunction with other hormones and endocrine systems. Its receptors are widely distributed across many tissues in the body, underscoring its multifaceted influence.
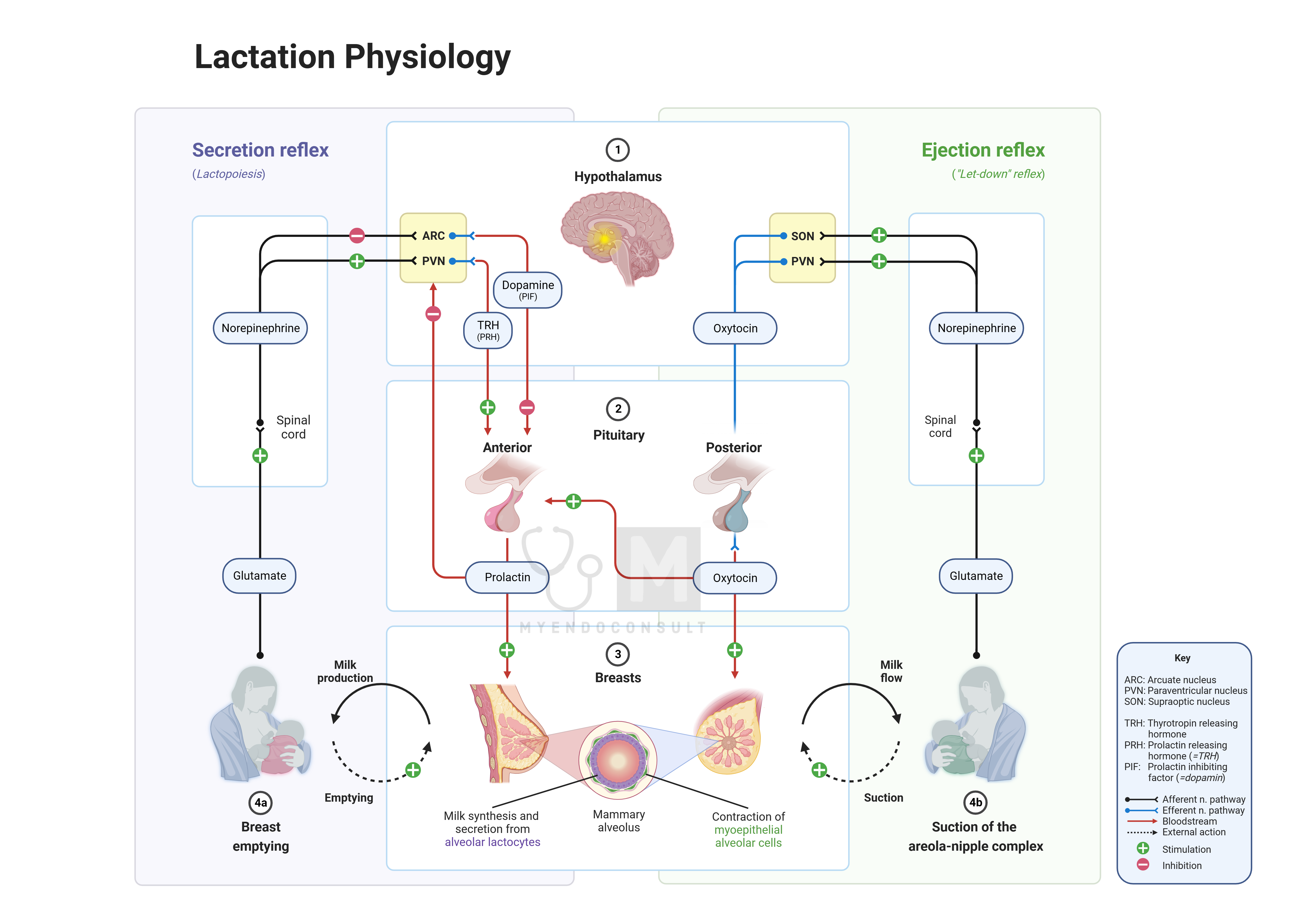
Lactation:
The most well-known and crucial physiological effect of prolactin is in lactogenesis – stimulating milk production in the mammary glands during lactation. This function is essential for the survival of infants in the early months of life, as it provides the primary source of nutrition. During pregnancy, high levels of estrogens and progesterone inhibit prolactin’s lactogenic effect. However, after childbirth, with the sharp decline in gonadal steroids, this inhibition is lifted, allowing prolactin to stimulate milk production. If breastfeeding is not initiated soon after birth and maintained regularly, prolactin production decreases, and its levels return to normal.
Breast Growth and Development:
Prolactin also contributes to the development and growth of the mammary gland during pregnancy, alongside hormones like estrogens, progesterone, growth hormone (GH), and locally produced insulin-like growth factor 1 (IGF1). The pituitary gland can enlarge significantly during pregnancy, primarily due to increased lactotrophs producing prolactin, leading to elevated plasma prolactin concentrations. Notably, the initial phase of mammary development during puberty is less dependent on prolactin and more on GH-mediated IGF1.
Immune System:
Prolactin’s role extends beyond gender-specific functions, as it is synthesized in both males and females and present in the circulation of both genders. This indicates potential non-gender-specific physiological effects, such as interactions with the immune system. Prolactin receptors are found in various tissues, including lymphoid tissues. Additionally, prolactin is produced not only by anterior pituitary lactotrophs but also by immune cells, including human lymphocytes. There is evidence suggesting that prolactin stimulates the proliferation of several cell types, including T cells, highlighting its significant role in the immune response.
Clinical presentation of prolactinomas
In women, prolactin-secreting microadenomas (tumors ≤10 mm in diameter) often present as secondary amenorrhea with or without galactorrhea. In men, and postmenopausal women, small prolactinomas usually go undiagnosed until they enlarge enough to cause mass-effect symptoms, such as visual field defects, cranial nerve palsies (e.g., diplopia, ptosis), headaches, and varying degrees of hypopituitarism.
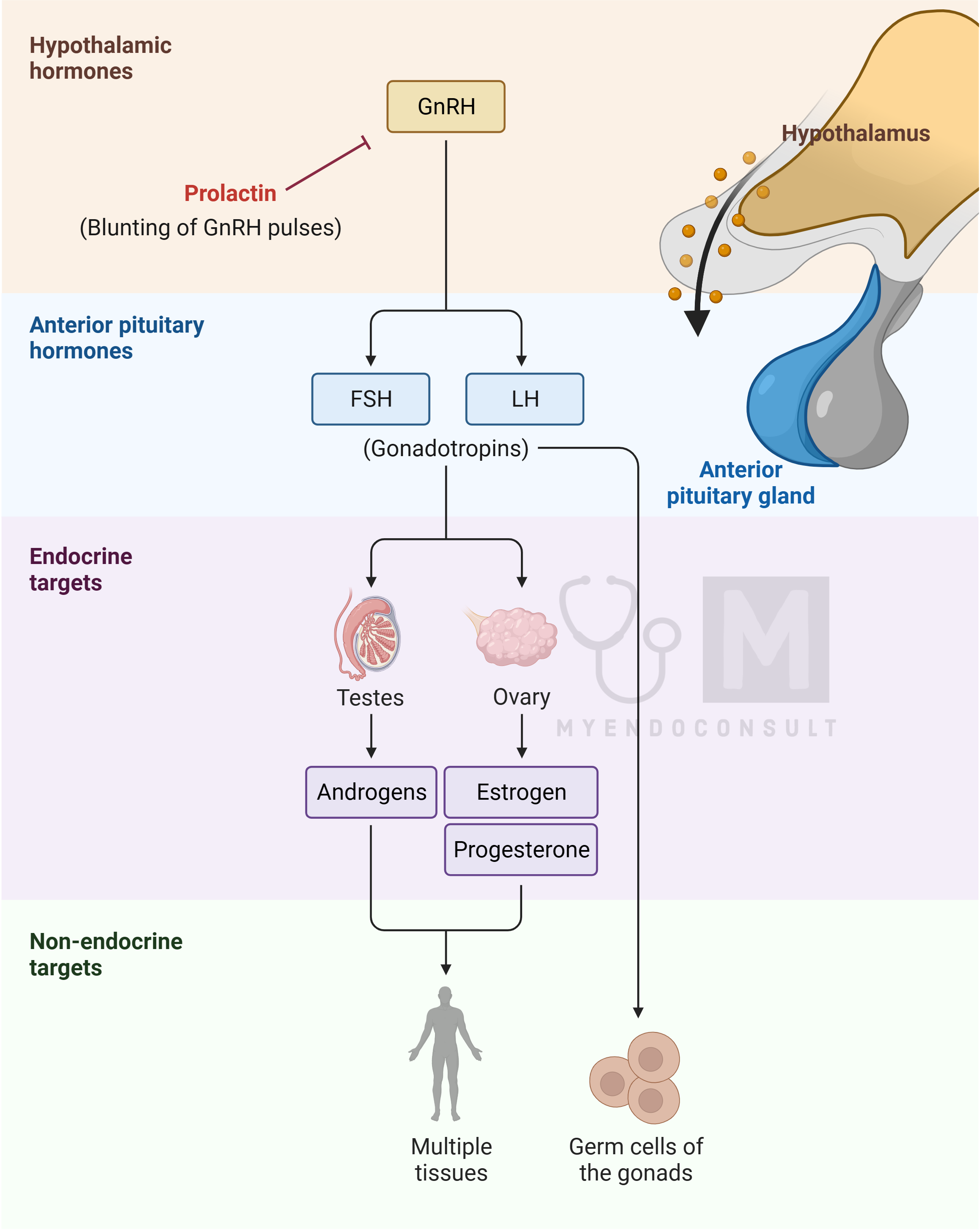
Hyperprolactinemia leads to decreased gonadotropin secretion in both men and women. Men experience hypogonadotropic hypogonadism, leading to symptoms like testicular atrophy, low testosterone, decreased libido, sexual dysfunction, reduced facial hair growth, and muscle mass loss. Galactorrhea is rare in men due to the lack of estrogen. In premenopausal women, hyperprolactinemia can cause bilateral spontaneous or expressible galactorrhea, secondary amenorrhea, and estrogen deficiency symptoms. Long-term hypogonadism in both genders may result in osteopenia and osteoporosis.
Evaluation of prolactinomas
The blood concentration of prolactin generally correlates with the size of the prolactinoma. For instance, a 5-mm prolactinoma might produce serum prolactin levels between 50 to 250 ng/mL, whereas larger tumors (>2 cm) often result in levels greater than 1000 ng/mL. However, some small prolactinomas may have high prolactin secretory capacity, and conversely, some large tumors may have inefficient prolactin secretion.
Treatment for prolactin-secreting pituitary tumors is based on the symptoms of hyperprolactinemia and mass-effect symptoms. Small, asymptomatic microadenomas in postmenopausal women might not require immediate treatment, while macroprolactinomas generally necessitate treatment due to their potential growth, regardless of symptom presence
Treatment of Prolactin-Secreting Pituitary Tumors (Prolactinomas)
When treatment for prolactinomas is indicated, such as in cases of secondary hypogonadism in men or premenopausal women, or if a macro adenoma is present, the preferred approach is medication with an orally administered dopamine agonist. Common choices include cabergoline or bromocriptine. These medications are highly effective in quickly normalizing serum prolactin levels and reducing the size of the lactotroph adenoma.
Once treatment with a dopamine agonist begins, it is crucial to monitor the serum prolactin concentration every 2 weeks. The dosage of the dopamine agonist should be adjusted to ensure prolactin levels fall within the reference range. After achieving normoprolactinemia, typically within 3 to 6 months, a pituitary-directed MRI should be conducted to confirm tumor shrinkage.
Patients should continue the minimal effective dosage of the dopamine agonist indefinitely. However, since a small percentage of patients might experience a cure from long-term dopamine agonist therapy, it is recommended to take a 2-month break from the medication every 2 years to assess if hyperprolactinemia recurs.
Patients with macroprolactinomas extending into the sphenoid sinus should be warned about the potential risk of cerebrospinal fluid (CSF) rhinorrhea as the tumor shrinks. CSF rhinorrhea is a serious condition requiring urgent neurosurgical intervention to prevent complications like pneumocephalus and bacterial meningitis.
In cases where patients are intolerant to dopamine agonists, experiencing side effects like nausea, lightheadedness, mental fogginess, or vivid dreams, or if the tumor is resistant to medication, alternative treatments like transsphenoidal surgery or gamma knife radiation therapy may be considered. These options are typically reserved for more challenging cases where medical therapy is insufficient or unsuitable.

